Bottom-up physiologically-based biokinetic modelling as an alternative to animal testing
Main Article Content
Abstract
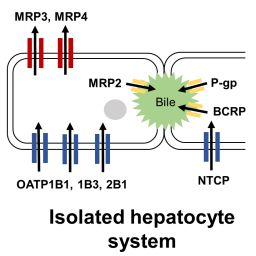
There is a growing need to develop alternatives to animal testing to derive biokinetic data for evaluating both efficacy and safety of chemicals. One such alternative is bottom-up physiologically-based biokinetic (PBK) modeling, which requires only in vitro data. The primary objective of this study was to develop and validate bottom-up PBK models of 3 HMG-CoA reductase inhibitors: rosuvastatin, fluvastatin, and pitavastatin. Bottom-up PBK models were built using the Simcyp® simulator by incorporating in vitro transporter and metabolism data (Vmax, Jmax, Km, CLint) obtained from the literature and proteomics-based scaling factors to account for differences in transporter expression between in vitro systems and in vivo organs. Simulations were performed for single intravenous, single oral, and multiple oral doses of these chemicals. The results showed that our bottom-up models predicted systemic exposure (AUC0h-t), maximum plasma concentration (Cmax), plasma clearance, and time to reach Cmax (Tmax) within two-fold of the observed data, with the exception of parameters associated with multiple oral pitavastatin dosing and single oral fluvastatin dosing. Additional middle-out simulations were performed using animal distribution data to inform tissue-to-plasma equilibrium distribution ratios for rosuvastatin and pitavastatin. This improved the predicted plasma-concentration time profiles but did not significantly alter the predicted biokinetic parameters. Our study demonstrates that quantitative proteomics-based mechanistic in vitro-to-in vivo extrapolation (IVIVE) can account for downregulation of transporters in culture and predict whole organ clearance without empirical scaling. Hence, bottom-up PBK modeling incorporating mechanistic IVIVE could be a viable alternative to animal testing in predicting human biokinetics.
Article Details
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).