The HaCaT/THP-1 cocultured activation test (COCAT) for skin sensitization: A study of intra-laboratory reproducibility and predictivity
Main Article Content
Abstract
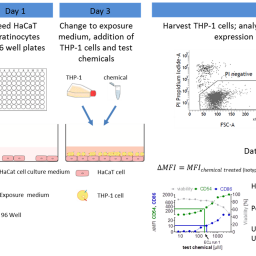
The Cocultured Activation Test (COCAT) consists of cocultured HaCaT (human keratinocyte cell line) and THP-1 cells (surrogate of antigen presenting cells). Individually, these cell lines are used to address key events 2 and 3 of the skin sensitization adverse outcome pathway (AOP). Their exposure in coculture was found to have the potential to increase their response to sensitizing chemicals, enable the detection of pro-haptens, and support the identification of skin sensitization potency. The present study was undertaken to assess the predictive capacity of COCAT of both skin sensitization hazard and potency and to assess the intra-laboratory reproducibility of COCAT based on the blind testing of chemicals. Results showed a reproducibility between runs of 80% for 15 coded chemicals. Skin sensitization hazard prediction had 100% sensitivity (9/9), 75% specificity (3/4), and 92.3% accuracy (12/13), while the tests of two chemicals were inconclusive. Including additional chemicals tested during the optimization phase in addition to the blind tested chemicals, the skin sensitization UN GHS sub-categories were correctly predicted for 85.7% (12/14) sub-category 1A chemicals, 83.3% (10/12) sub-category 1B chemicals, and 92.3% (12/13) no category chemicals, resulting in an overall accuracy of 87.4% (34/39). The present study shows the COCAT to be a promising method for the identification of skin sensitization hazard and potency sub-categorization according to the UN GHS classification.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).