Developing context appropriate toxicity testing approaches using new alternative methods (NAMs)
Main Article Content
Abstract
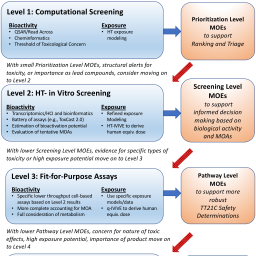
In the past 10 years, the public, private, and non-profit sectors have found agreement that hazard identification and risk assessment should capitalize on the explosion of knowledge in the biological sciences, moving away from in life animal testing toward more human-relevant in vitro and in silico methods, collectively referred to as new approach methodologies (NAMs). The goals for implementation of NAMs are to efficiently identify possible chemical hazards and to gather dose-response data to inform more human-relevant safety assessment. While work proceeds to develop NAMs, there has been less emphasis on creating decision criteria or showing how risk context should guide selection and use of NAMs. Here, we outline application scenarios for NAMs in different risk contexts and place different NAMs and conventional testing approaches into four broad levels. Level 1 relies solely on computational screening; Level 2 consists of high throughput in vitro screening with human cells intended to provide broad coverage of possible responses; Level 3 focuses on fit-for-purpose assays selected based on presumptive modes of action (MOA) and designed to provide more quantitative estimates of relevant dose responses; Level 4 has a variety of more complex multi-dimensional or multi-cellular assays and might include targeted in vivo studies to further define MOA. Each level also includes decision-appropriate exposure assessment tools. Our aims here are to (1) foster discussion about context-dependent applications of NAMs in relation to risk assessment needs and (2) describe a functional roadmap to identify where NAMs are expected to be adequate for chemical safety decision-making.
Article Details
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).