Optimization of the monocyte activation test for evaluating pyrogenicity of tick-borne encephalitis virus vaccine
Main Article Content
Abstract
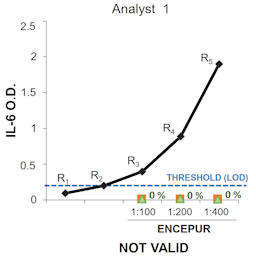
Pyrogen content is a key quality feature that must be checked in all injectable products, including vaccines. Four tests are currently available in the European Pharmacopoeia to monitor pyrogen/endotoxin presence: the rabbit pyrogen test (RPT), the bacterial endotoxin test, the recombinant factor C test, and the monocyte activation test (MAT). Here, we explored the possibility to replace the RPT with the MAT in the quality control of a vaccine against tick-borne encephalitis virus (TBEV). The testing was carried out using cryopreserved peripheral blood mononuclear cells as cell source. IL-6 release was selected as readout for the detection of both endotoxin and non-endotoxin contaminants. MAT applicability for pyrogen testing of the TBEV vaccine was assessed through preparatory tests and resulted in the establishment of a very sensitive assay (limit of detection (LOD) = 0.04 EU/mL; sensitivity = 0.1 EU/mL). Both quantitative Method A and semiquantitative Method B were used for data analysis. Our studies revealed that for a vaccine without intrinsic pyrogenicity, such as that against TBEV, sensitivity (the lowest endotoxin value of the standard curve) should be used instead of LOD to define a stable maximum valid dilution of the product. In conclusion, we describe the challenges of MAT implementation for anti-TBEV vaccine following the current Ph. Eur. chapter 2.6.30 and propose a re-evaluation of the validity criteria of Methods A and B in order to set a semi-quantitative or limit test suitable for those products for which a reference lot comparison analysis is not applicable or favorable.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).