Galleria mellonella as an alternative in vivo model to study bacterial biofilms on stainless steel and titanium implants
Main Article Content
Abstract
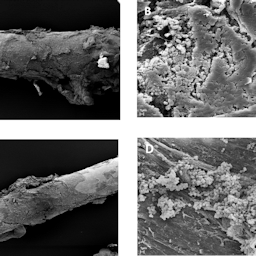
The purpose of this study was to establish an infection model of Galleria mellonella larvae as an alternative in vivo model for biofilm-associated infections on stainless steel and titanium implants. First, the model was established with sterile implants to evaluate biocompatibility. Titanium or stainless steel implants were implanted without adverse effects over the entire observation period of 5 days compared to controls and even up to the pupae and moth stage. Then, stainless steel and titanium implants contaminated with Staphylococcus aureus were implanted into larvae to mimic biofilm-associated infection. For both materials, pre-incubation of the implant with S. aureus led to significantly reduced survival of the larvae compared to sterile implants. Larvae could not be rescued by gentamicin, whereas gentamicin significantly improved the survival of the larvae in case of planktonic infection with S. aureus without an implant, confirming the typical characteristics of reduced antibiotic susceptibility of biofilm infections. Biofilm formation and various stages of biofilm maturation were confirmed by surface electron microscopy and by measuring bacterial gene expression of biofilm-related genes on contaminated implants, which confirmed biofilm formation and upregulation of autolysin (atl ) and sarA genes. In conclusion, G. mellonella can be used as an alternative in vivo model to study biofilm-associated infections on stainless steel and titanium implants, which may help to reduce animal infection experiments with vertebrates in the future.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Alt, V., Lips, K. S., Henkenbehrens, C. et al. (2011). A new animal model for implant-related infected non-unions after intramedullary fixation of the tibia in rats with fluorescent in situ hybridization of bacteria in bone infection. Bone 48, 1146-1153. doi:10.1016/j.bone.2011.01.018
Azmi, K., Qrei, W. and Abdeen, Z. (2019). Screening of genes encoding adhesion factors and biofilm production in methicillin resistant strains of Staphylococcus aureus isolated from Palestinian patients. BMC Genomics 20, 578. doi:10.1186/s12864-019-5929-1
Balamurugan, P., Praveen Krishna, V., Bharath, D. et al. (2017). Staphylococcus aureus quorum regulator SarA targeted compound, 2-[(Methylamino)methyl]phenol inhibits biofilm and down-regulates virulence genes. Front Microbiol 8, 1290. doi:10.3389/fmicb.2017.01290
Bechert, T., Steinrücke, P. and Guggenbichler, J. P. (2000). A new method for screening anti-infective biomaterials. Nat Med 6, 1053-1056. doi:10.1038/79568
Beenken, K. E., Blevins, J. S. and Smeltzer, M. S. (2003). Mutation of sarA in Staphylococcus aureus limits biofilm formation. Infect Immun 71, 4206-4211. doi:10.1128/IAI.71.7.4206-4211.2003
Benthall, G., Touzel, R. E., Hind, C. K. et al. (2015). Evaluation of antibiotic efficacy against infections caused by planktonic or biofilm cultures of Pseudomonas aeruginosa and Klebsiella pneumoniae in Galleria mellonella. Int J Antimicrob Agents 46, 538-545. doi:10.1016/j.ijantimicag.2015.07.014
Campos-Silva, R., Brust, F. R., Trentin, D. S. et al. (2019). Alternative method in Galleria mellonella larvae to study biofilm infection and treatment. Microb Pathog 137, 103756. doi:10.1016/j.micpath.2019.103756
Dai, L., Yang, L., Parsons, C. et al. (2012). Staphylococcus epidermidis recovered from indwelling catheters exhibit enhanced biofilm dispersal and “self-renewal” through downregulation of agr. BMC Microbiol 12, 102. doi:10.1186/1471-2180-12-102
Joo, H. S. and Otto, M. (2012). Molecular basis of in vivo biofilm formation by bacterial pathogens. Chem Biol 19, 1503-1513. doi:10.1016/j.chembiol.2012.10.022
Khatoon, Z., McTiernan, C. D., Suuronen, E. J. et al. (2018). Bacterial biofilm formation on implantable devices and approaches to its treatment and prevention. Heliyon 4, e01067. doi:10.1016/j.heliyon.2018.e01067
Kot, B., Sytykiewicz, H. and Sprawka, I. (2018). Expression of the biofilm-associated genes in methicillin-resistant Staphylococcus aureus in biofilm and planktonic conditions. Int J Mol Sci 19, 3487. doi:10.3390/ijms19113487
Le, K. Y. and Otto, M. (2015). Quorum-sensing regulation in staphylococci – An overview. Front Microbiol 6, 1174. doi:10.3389/fmicb.2015.01174
Lebeaux, D., Chauhan, A., Rendueles, O. et al. (2013). From in vitro to in vivo models of bacterial biofilm-related infections. Pathogens 2, 288-356. doi:10.3390/pathogens2020288
Mannala, G. K., Izar, B., Rupp, O. et al. (2017a). Listeria monocytogenes induces a virulence-dependent microRNA signature that regulates the immune response in Galleria mellonella. Front Microbiol 8, 2463. doi:10.3389/fmicb.2017.02463
Mannala, G. K., Hain, T., Spröer, C. et al. (2017b). Complete genome and plasmid sequences of Staphylococcus aureus EDCC 5055 (DSM 28763), used to study implant-associated infections. Genome Announc 5, e01698-16. doi:10.1128/genomeA.01698-16
Mannala, G. K., Koettnitz, J., Mohamed, W. et al. (2018). Whole-genome comparison of high and low virulent Staphylococcus aureus isolates inducing implant-associated bone infections. Int J Med Microbiol 308, 505-513. doi:10.1016/j.ijmm.2018.04.005
Moriarty, T. F., Schmid, T., Post, V. et al. (2017). A large animal model for a failed two-stage revision of intramedullary nail-related infection by methicillin-resistant Staphylococcus aureus. Eur Cells Mater 34, 83-98. doi:10.22203/eCM.v034a06
Moriarty, T. F., Harris, L. G., Mooney, R. A. et al. (2019). Recommendations for design and conduct of preclinical in vivo studies of orthopedic device-related infection. J Orthop Res 37, 271-287. doi:10.1002/jor.24230
Mukherjee, K., Altincicek, B., Hain, T. et al. (2010). Galleria mellonella as a model system for studying Listeria pathogenesis. Appl Environ Microbiol 76, 310-317. doi:10.1128/AEM.01301-09
Nishitani, K., Sutipornpalangkul, W., De Mesy Bentley, K. L. et al. (2015). Quantifying the natural history of biofilm formation in vivo during the establishment of chronic implant-associated Staphylococcus aureus osteomyelitis in mice to identify critical pathogen and host factors. J Orthop Res 33, 1311-1319. doi:10.1002/jor.22907
Paharik, A. E. and Horswill, A. R. (2016). The Staphylococcal biofilm: Adhesins, regulation, and host response. Microbiol Spectr 4. doi:10.1128/microbiolspec.vmbf-0022-2015
Pfaffl, M. W. (2001). A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29, e45. doi:10.1093/nar/29.9.e45
Ribeiro, M., Monteiro, F. J. and Ferraz, M. P. (2012). Infection of orthopedic implants with emphasis on bacterial adhesion process and techniques used in studying bacterial-material interactions. Biomatter 2, 176-194. doi:10.4161/biom.22905
Russell, W. M. S. and Burch, R. L. (1959). The Principles of Humane Experimental Technique. https://caat.jhsph.edu/principles/the-principles-of-humane-experimental-technique
Tsang, L. H., Cassat, J. E., Shaw, L. N. et al. (2008). Factors contributing to the biofilm-deficient phenotype of Staphylococcus aureus sarA mutants. PLoS One 3, e3361. doi:10.1371/journal.pone.0003361
Valle, J., Vergara-Irigaray, M., Merino, N. et al. (2007). sigmaB regulates IS256-mediated Staphylococcus aureus biofilm phenotypic variation. J Bacteriol 189, 2886-2896. doi:10.1128/JB.01767-06
Waldvogel, F. A., Bisno, A. L. (eds.) (2000). Infections Associated with Indwelling Medical Devices, Third Edition. American Society of Microbiology. doi:10.1128/9781555818067
Yarwood, J. M., Bartels, D. J., Volper, E. M. et al. (2004). Quorum sensing in Staphylococcus aureus biofilms. J Bacteriol 186, 1838-1850. doi:10.1128/JB.186.6.1838-1850.2004
Zimmerli, W., Trampuz, A. and Ochsner, P. E. (2004). Prosthetic-joint infections. N Engl J Med 351, 1645-1654. doi:10.1056/NEJMra040181