Tradition, not science, is the basis of animal model selection in translational and applied research
Main Article Content
Abstract
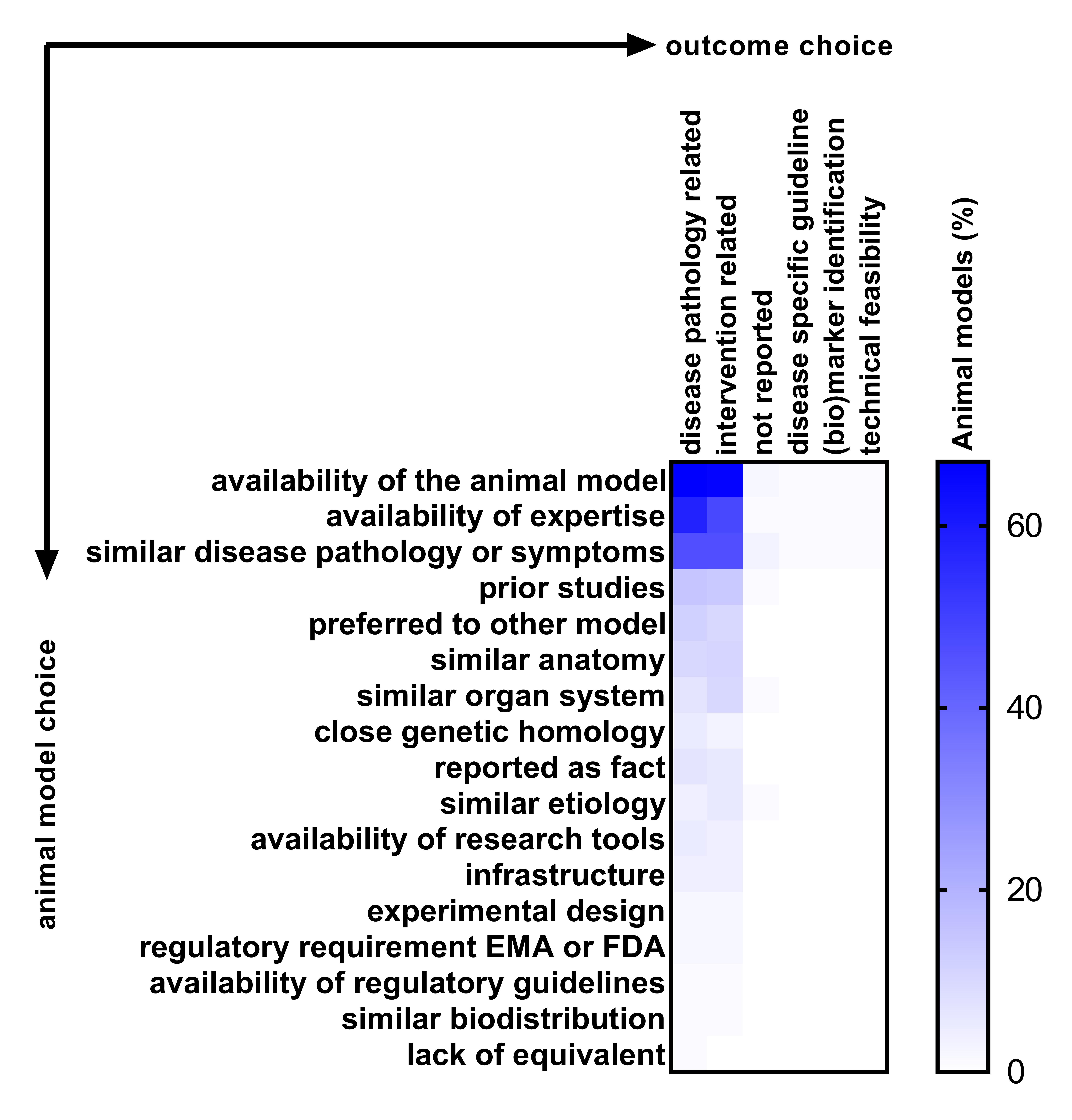
National and international laws and regulations exist to protect animals used for scientific purposes in translational and applied research, which includes drug development. However, multiple animal models are available for each disease. We evaluated the argumentation behind the selection of a specific animal model using thematic content analysis in project applications issued in 2017-2019 in the Netherlands. In total, 125 animal models for translational and applied research from 110 project applications were assessed. Explanations to select a specific model included: the model’s availability (79%); the availability of expertise (62%); and the model showing similar disease pathology/symptoms (59%) to humans. Therefore, current selection of a specific animal model seems to be based on tradition rather than its potential predictive value for clinical outcome. The applicants’ explanations for the implementation of the 3R principles (replacement, reduction and refinement) as to the animal model were unspecific. Replacement was achieved by using data from prior in vitro studies, reduction by optimal experimental design and statistics, and refinement by reducing discomfort. Additionally, due to the stated need for a test model with high complexity (47%) and intactness (30%), the full replacement of animal models with alternative (non-live animal) approaches was thought unachievable. Without a clear, systematic and transparent justification for the selection of a specific animal model, the likelihood of poorly translatable research remains. It is not only up to the researcher to demonstrate this, as ethical committees and funding bodies can provide positive stimuli to drive this change.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Binnenlandse Zaken en Koninkrijksrelaties (2018). Wet openbaarheid van bestuur. Staatsblad van het Koninkrijk der Nederlanden 248, BWBR0005252.
Denayer, T., Stöhr, T. and Van Roy, M. (2014). Animal models in translational medicine: Validation and prediction. New Horiz Transl Med 2, 5-11. doi:10.1016/j.nhtm.2014.08.001
Elo, S. and Kyngas, H. (2008). The qualitative content analysis process. J Adv Nurs 62, 107-115. doi:10.1111/j.1365-2648.2007.04569.x
EC – European Commission (2012). Commission Implementing Decision of 14 November 2012 establishing a common format for the submission of the information pursuant to Directive 2010/63/EU of the European Parliament and of the Council on the protection of animals used for scientific purposes. OJ L 320, 33-50. http://data.europa.eu/eli/dec_impl/2012/707/oj
EC (2020). Commission Implementing Decision (EU) 2020/569 of 16 April 2020 establishing a common format and information content for the submission of the information to be reported by member states pursuant to Directive 2010/63/EU of the European Parliament and of the Council on the protection of animals used for scientific purposes and repealing Commission Implementing Decision 2012/707/EU (notified under document C(2020) 2179). OJ L 129, 16-50. http://data.europa.eu/eli/dec_impl/2020/569/oj
EU – European Union (2010). Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes. OJ L 276, 33-79. http://data.europa.eu/eli/dir/2010/63/oj
Ferreira, G. S., Veening-Griffioen, D. H., Boon, W. P. C. et al. (2019). A standardised framework to identify optimal animal models for efficacy assessment in drug development. PLoS One 14, e0218014. Erratum in: PLoS One 14, e0220325. doi:10.1371/journal.pone.0218014
Fontanarosa, P. B. and DeAngelis, C. D. (2003). Translational medical research. JAMA 289, 2133. doi:10.1001/jama.289.16.2133
Freedman, L. P., Cockburn, I. M. and Simcoe, T. S. (2015). The economics of reproducibility in preclinical research. PLoS Biol 13, e1002165. doi:10.1371/journal.pbio.1002165
Godlee, F. (2018). We need better animal research, better reported. BMJ 360, k124. doi:10.1136/bmj.k124
Harrison, R. K. (2016). Phase II and phase III failures: 2013-2015. Nat Rev Drug Discov 15, 817-818. doi:10.1038/nrd.2016.184
Hau, J. (2008). Animal models for human diseases. In P. M. Conn (ed.), Sourcebook of Models for Biomedical Research. Totowa, NJ, USA: Humana Press. doi:10.1007/978-1-59745-285-4_1
Held, J. R. (1980). Muhlbock memorial lecture: Considerations in the provision and characterization of animal models. In A. Spiegel, S. Erichsen and H. A. Solleveld (eds.), Animal Quality and Models in Biomedical Research, 7th ICLAS Symposium Utrecht 1979. Stuttgart, Germany: Gustav Fisher Verlag.
Herrmann, K. and Flecknell, P. (2018). The application of humane endpoints and humane killing methods in animal research proposals: A retrospective review. Altern Lab Anim 46, 317-333. doi:10.1177/026119291804600606
Herrmann, K. and Flecknell, P. (2019). Retrospective review of anesthetic and analgesic regimens used in animal research proposals. ALTEX 36, 65-80. doi:10.14573/altex.1804011
Kilkenny, C., Browne, W. J., Cuthill, I. C. et al. (2010). Improving bioscience research reporting: The ARRIVE guidelines for reporting animal research. PLoS Biol 8, e1000412. doi:10.1371/journal.pbio.1000412
Kooijman, M. (2013). Why animal studies are still being used in drug development. Altern Lab Anim 41, P79-81. doi:10.1177/026119291304100627
Langhof, H., Chin, W. W. L., Wieschowski, S. et al. (2018). Preclinical efficacy in therapeutic area guidelines from the U.S. Food and Drug Administration and the European Medicines Agency: A cross-sectional study. Br J Pharmacol 175, 4229-4238. doi:10.1111/bph.14485
Leaman, J., Latter, J. and Clemence, M. (2014). Attitudes to animal research in 2014. Ipsos MORI Social Research Institute, London. https://www.ipsos.com/sites/default/files/migrations/en-uk/files/Assets/Docs/Polls/sri_BISanimalresearch_NONTRENDreport.pdf (accessed 13.12.2019)
Leung, V., Rousseau-Blass, F., Beauchamp, G. et al. (2018). ARRIVE has not ARRIVEd: Support for the ARRIVE (animal research: Reporting of in vivo experiments) guidelines does not improve the reporting quality of papers in animal welfare, analgesia or anesthesia. PLoS One 13, e0197882. doi:10.1371/journal.pone.0197882
Macleod, M. and Mohan, S. (2019). Reproducibility and rigor in animal-based research. ILAR J 60, 17-23. doi:10.1093/ilar/ilz015
Nederlandse Voedsel- en Warenautoriteit (2018). Zo doende 2017. Jaaroverzicht dierproeven en proefdieren van de nederlande voedsel- en warenautoriteit. Nederlandse Voedsel- en Warenautoriteit, 1-62.
Percie du Sert, N., Hurst, V., Ahluwalia, A. et al. (2019). The ARRIVE guidelines 2019: Updated guidelines for reporting animal research. PLoS Biol 18, e3000410. doi:10.1371/journal.pbio.3000410
Pound, P., Ebrahim, S., Sandercock, P. et al. (2004). Where is the evidence that animal research benefits humans? BMJ 328, 514-517. doi:10.1136/bmj.328.7438.514
Pound, P. and Blaug, R. (2016). Transparency and public involvement in animal research. Altern Lab Anim 44, 167-173. doi:10.1177/026119291604400210
Schulz, J. B., Cookson, M. R. and Hausmann, L. (2016). The impact of fraudulent and irreproducible data to the translational research crisis – Solutions and implementation. J Neurochem 139, Suppl 2, 253-270. doi:10.1111/jnc.13844
Smith, A. J., Clutton, R. E., Lilley, E. et al. (2018). Prepare: Guidelines for planning animal research and testing. Lab Anim 52, 135-141. doi:10.1177/0023677217724823
van der Worp, H. B., Howells, D. W., Sena, E. S. et al. (2010). Can animal models of disease reliably inform human studies? PLoS Med 7, e1000245. doi:10.1371/journal.pmed.1000245
Veening-Griffioen, D. H., Ferreira, G. S., van Meer, P. J. K. et al. (2019). Are some animal models more equal than others? A case study on the translational value of animal models of efficacy for Alzheimer’s disease. Eur J Pharmacol 859, 172524. doi:10.1016/j.ejphar.2019.172524
Woolf, S. H. (2008). The meaning of translational research and why it matters. JAMA 299, 211-213. doi:10.1001/jama.2007.26