Limitations of animal epilepsy research models: Can epileptic human tissue provide translational benefit?
Main Article Content
Abstract
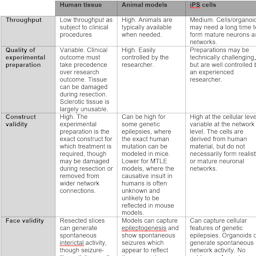
Advancement of understanding the etiology and treatment of epilepsy has largely depended on the use of acute and chronic animal models. An alternative approach, which is being increasingly used by a select number of laboratories worldwide, is to perform functional mechanistic studies in brain slices of living human tissue resected during surgery for drug resistant epilepsies. Pharmacoresistant epilepsy is a major clinical problem with a significant proportion of patients not receiving any symptomatic benefit from available anti-epileptic drugs. Animal models of epilepsy have dominated the landscape with regard to research and development, however they have failed to deliver new agents that would provide seizure control in patients with drug refractory epilepsy. Moreover, these models have considerable issues with respect to validity and animal welfare considerations. A compelling alternative is the use of live human epileptic tissue, which recapitulates a number of key features of refractory epilepsy. The use of live epileptic human tissue offers unprecedented opportunities to understand the mechanisms associated with difficult to treat epilepsy whilst also permitting studies of efficacy of novel agents that are being developed to alleviate epilepsy in drug resistant patients.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Akiyama, T., McCoy, B., Go, C. Y. et al. (2011). Focal resection of fast ripples on extraoperative intracranial EEG improves seizure outcome in pediatric epilepsy. Epilepsia 52, 1802-1811. doi:10.1111/j.1528-1167.2011.03199.x
Annegers, J. F., Hauser, W. A., Shirts, S. B. et al. (1987). Factors prognostic of unprovoked seizures after febrile convulsions. N Engl J Med 316, 493-498. doi:10.1056/nejm198702263160901
Aronica, E., Gorter, J. A., Ramkema, M. et al. (2004). Expression and cellular distribution of multidrug resistance-related proteins in the hippocampus of patients with mesial temporal lobe epilepsy. Epilepsia 45, 441-451. doi:10.1111/j.0013-9580.2004.57703.x
Aronica, E., Sisodiya, S. M. and Gorter, J. A. (2012). Cerebral expression of drug transporters in epilepsy. Adv Drug Deliv Rev 64, 919-929. doi:10.1016/j.addr.2011.11.008
Ben-Ari, Y. and Lagowska, J. (1978). Epileptogenic action of intra-amygdaloid injection of kainic acid. C R Acad Sci Hebd Seances Acad Sci D 287, 813-816 [Article in French]. http://www.ncbi.nlm.nih.gov/pubmed/103652
Ben-Ari, Y., Lagowska, J., Tremblay, E. et al. (1979). A new model of focal status epilepticus: Intra-amygdaloid application of kainic acid elicits repetitive secondarily generalized convulsive seizures. Brain Res 163, 176-179. doi:10.1016/0006-8993(79)90163-X
Ben-Ari, Y., Tremblay, E., Ottersen, O. P. et al. (1980). The role of epileptic activity in hippocampal and ‘remote’ cerebral lesions induced by kainic acid. Brain Res 191, 79-97. doi:10.1016/0006-8993(80)90316-9
Benardo, L. S. (2003). Altered sodium channels underlie anticonvulsant drug insensitivity. Epilepsy Curr 3, 227-228. doi:10.1046/j.1535-7597.2003.03606.x
Benito-Kwiecinski, S. and Lancaster, M. A. (2019). Brain organoids: Human neurodevelopment in a dish. Cold Spring Harb Perspect Biol, a035709. doi:10.1101/cshperspect.a035709
Blümcke, I. (2009). Neuropathology of focal epilepsies: A critical review. Epilepsy Behav 15, 34-39. doi:10.1016/j.yebeh.2009.02.033
Blümcke, I., Thom, M., Aronica, E. et al. (2013). International consensus classification of hippocampal sclerosis in temporal lobe epilepsy: A task force report from the ILAE commission on diagnostic methods. Epilepsia 54, 1315-1329. doi:10.1111/epi.12220
Brandt, C., Glien, M., Potschka, H. et al. (2003). Epileptogenesis and neuropathology after different types of status epilepticus induced by prolonged electrical stimulation of the basolateral amygdala in rats. Epilepsy Res 55, 83-103. doi:10.1016/S0920-1211(03)00114-1
Brandt, C., Volk, H. A. and Loscher, W. (2004). Striking differences in individual anticonvulsant response to phenobarbital in rats with spontaneous seizures after status epilepticus. Epilepsia 45, 1488-1497. doi:10.1111/j.0013-9580.2004.16904.x
Buskila, Y., Breen, P. P., Tapson, J. et al. (2014). Extending the viability of acute brain slices. Sci Rep 4, 4-10. doi:10.1038/srep05309
Cho, J. R., Joo, E. Y., Koo, D. L. et al. (2012). Clinical utility of interictal high-frequency oscillations recorded with subdural macroelectrodes in partial epilepsy. J Clin Neurol 8, 22. doi:10.3988/jcn.2012.8.1.22
Claes, L., Del-Favero, J., Ceulemans, B. et al. (2001). De novo mutations in the sodium-channel gene SCN1A cause severe myoclonic epilepsy of infancy. Am J Hum Genet 68, 1327-1332. doi:10.1086/320609
Claes, L., Ceulemans, B., Audenaert, D. et al. (2003). De novo SCN1A mutations are a major cause of severe myoclonic epilepsy of infancy. Hum Mutat 21, 615-621. doi:10.1002/humu.10217
Covolan, L. and Mello, L. E. A. (2000). Temporal profile of neuronal injury following pilocarpine or kainic acid-induced status epilepticus. Epilepsy Res 39, 133-152. doi:10.1016/S0920-1211(99)00119-9
Cunningham, M. O., Roopun, A., Schofield, I. S. et al. (2012). Glissandi: Transient fast electrocorticographic oscillations of steadily increasing frequency, explained by temporally increasing gap junction conductance. Epilepsia 53, 1205-1214. doi:10.1111/j.1528-1167.2012.03530.x
Curia, G., Longo, D., Biagini, G. et al. (2008). The pilocarpine model of temporal lobe epilepsy. J Neurosci Methods 172, 143-157. doi:10.1016/j.jneumeth.2008.04.019
de Tisi, J., Bell, G. S., Peacock, J. L. et al. (2011). The long-term outcome of adult epilepsy surgery, patterns of seizure remission, and relapse: A cohort study. Lancet 378, 1388-1395. doi:10.1016/S0140-6736(11)60890-8
Devinsky, O., Vezzani, A., Najjar, S. et al. (2013). Glia and epilepsy: Excitability and inflammation. Trends Neurosci 36, 174-184. doi:10.1016/j.tins.2012.11.008
Dodson, P. D. and Forsythe, I. D. (2004). Presynaptic K+ channels: Electrifying regulators of synaptic terminal excitability. Trends Neurosci 27, 210-217. doi:10.1016/j.tins.2004.02.012
Dubé, C., Richichi, C., Bender, R. A. et al. (2006). Temporal lobe epilepsy after experimental prolonged febrile seizures: Prospective analysis. Brain 129, 911-922. doi:10.1093/brain/awl018
Endele, S., Rosenberger, G., Geider, K. et al. (2010). Mutations in GRIN2A and GRIN2B encoding regulatory subunits of NMDA receptors cause variable neurodevelopmental phenotypes. Nat Genet 42, 1021-1026. doi:10.1038/ng.677
Engel, J. (1996). Introduction to temporal lobe epilepsy. Epilepsy Res 26, 141-150. doi:10.1016/s0920-1211(96)00043-5
Engel, J. (2001). Mesial temporal lobe epilepsy: What have we learned? Neurosci 7, 340-352. doi:10.1177/107385840100700410
Engel, J., Wiebe, S., French, J. et al. (2003). Practice parameter: Temporal lobe and localized neocortical resections for epilepsy. Epilepsia 44, 741-751. doi:10.1046/j.1528-1157.2003.48202.x
Engel, J. (2012). Early surgical therapy for drug-resistant temporal lobe epilepsy. JAMA 307, 922. doi:10.1001/jama.2012.220
Fang, M., Xi, Z.-Q., Wu, Y. et al. (2011). A new hypothesis of drug refractory epilepsy: Neural network hypothesis. Med Hypotheses 76, 871-876. doi:10.1016/j.mehy.2011.02.039
Flecknell, P. (2002). Replacement, reduction and refinement. ALTEX 19, 73-78. https://www.altex.org/index.php/altex/article/view/1106
French, E. D., Aldinio, C. and Schwarcz, R. (1982). Intrahippocampal kainic acid, seizures and local neuronal degeneration: Relationships assessed in unanesthetized rats. Neuroscience 7, 2525-2536. doi:10.1016/0306-4522(82)90212-3
French, J. A., Williamson, P. D., Thadani, V. M. et al. (1993). Characteristics of medial temporal lobe epilepsy: I. Results of history and physical examination. Ann Neurol 34, 774-780. doi:10.1002/ana.410340604
Glien, M., Brandt, C., Potschka, H. et al. (2002). Effects of the novel antiepileptic drug levetiracetam on spontaneous recurrent seizures in the rat pilocarpine model of temporal lobe epilepsy. Epilepsia 43, 350-357. doi:10.1046/j.1528-1157.2002.18101.x
Goldberg, E. M. and Coulter, D. A. (2013). Mechanisms of epileptogenesis: A convergence on neural circuit dysfunction. Nat Rev Neurosci 14, 337-349. doi:10.1038/nrn3482
Gribkoff, V. K. and Kaczmarek, L. K. (2017). The need for new approaches in CNS drug discovery: Why drugs have failed, and what can be done to improve outcomes. Neuropharmacology 120, 11-19. doi:10.1016/j.neuropharm.2016.03.021
Grone, B. P. and Baraban, S. C. (2015). Animal models in epilepsy research: Legacies and new directions. Nat Neurosci 18, 339-343. doi:10.1038/nn.3934
Haegelen, C., Perucca, P., Châtillon, C.-E. et al. (2013). High-frequency oscillations, extent of surgical resection, and surgical outcome in drug-resistant focal epilepsy. Epilepsia 54, 848-857. doi:10.1111/epi.12075
Jacobs, J., Zijlmans, M., Zelmann, R. et al. (2010). High-frequency electroencephalographic oscillations correlate with outcome of epilepsy surgery. Ann Neurol 67, 209-220. doi:10.1002/ana.21847
Jandová, K., Pasler, D., Antonio, L. L. et al. (2006). Carbamazepine-resistance in the epileptic dentate gyrus of human hippocampal slices. Brain 129, 3290-3306. doi:10.1093/brain/awl218
Jones, R. S. G., da Silva, A. B., Whittaker, R. G. et al. (2016). Human brain slices for epilepsy research: Pitfalls, solutions and future challenges. J Neurosci Methods 260, 221-232. doi:10.1016/j.jneumeth.2015.09.021
Köhling, R., Lücke, A., Straub, H. et al. (1998). Spontaneous sharp waves in human neocortical slices excised from epileptic patients. Brain 121, 1073-1087. doi:10.1093/brain/121.6.1073
Kwan, P., Arzimanoglou, A., Berg, A. T. et al. (2009). Definition of drug resistant epilepsy: Consensus proposal by the ad hoc task force of the ILAE commission on therapeutic strategies. Epilepsia 51, 1069-1077. doi:10.1111/j.1528-1167.2009.02397.x
LaRoche, S. M. (2007). A new look at the second-generation antiepileptic drugs: A decade of experience. Neurologist 13, 133-139. doi:10.1097/01.nrl.0000256353.14257.7c
Lévesque, M. and Avoli, M. (2013). The kainic acid model of temporal lobe epilepsy. Neurosci Biobehav Rev 37, 2887-2899. doi:10.1016/j.neubiorev.2013.10.011
Lidster, K., Jefferys, J. G., Blümcke, I. et al. (2016). Opportunities for improving animal welfare in rodent models of epilepsy and seizures. J Neurosci Methods 260, 2-25. doi:10.1016/j.jneumeth.2015.09.007
Liou, J., Smith, E. H., Bateman, L. M. et al. (2020). A model for focal seizure onset, propagation, evolution, and progression. Elife 9, e50927. doi:10.7554/eLife.50927
Löscher, W. (1997). Animal models of intractable epilepsy. Prog Neurobiol 53, 239-258. doi:10.1016/S0301-0082(97)00035-X
Löscher, W. (2002). Animal models of drug-resistant epilepsy. Novartis Found Symp 243, 149-159; discussion 159-166, 180-185.
Löscher, W. (2011). Critical review of current animal models of seizures and epilepsy used in the discovery and development of new antiepileptic drugs. Seizure 20, 359-368. doi:10.1016/j.seizure.2011.01.003
Lytton, W. W. (2008). Computer modelling of epilepsy. Nat Rev Neurosci 9, 626-637. doi:10.1038/nrn2416
Maglóczky, Z. (2010). Sprouting in human temporal lobe epilepsy: Excitatory pathways and axons of interneurons. Epilepsy Res 89, 52-59. doi:10.1016/j.eplepsyres.2010.01.002
Marwick, K., Skehel, P., Hardingham, G. et al. (2015). Effect of a GRIN2A de novo mutation associated with epilepsy and intellectual disability on NMDA receptor currents and Mg2+ block in cultured primary cortical neurons. Lancet 385, Suppl 1, S65. doi:10.1016/S0140-6736(15)60380-4
Mathern, G. W., Pretorius, J. K. and Babb, T. L. (1995). Influence of the type of initial precipitating injury and at what age it occurs on course and outcome in patients with temporal lobe seizures. J Neurosurg 82, 220-227. doi:10.3171/jns.1995.82.2.0220
Mathern, G., Bertram, E., Babb, T. et al. (1997). In contrast to kindled seizures, the frequency of spontaneous epilepsy in the limbic status model correlates with greater aberrant fascia dentata excitatory and inhibitory axon sprouting, and increased staining for N-methyl-d-aspartate, AMPA and GABAA rec. Neuroscience 77, 1003-1019. doi:10.1016/S0306-4522(96)00516-7
Mellanby, J., George, G., Robinson, A. et al. (1977). Epileptiform syndrome in rats produced by injecting tetanus toxin into the hippocampus. J Neurol Neurosurg Psychiatry 40, 404-414. doi:10.1136/jnnp.40.4.404
Mello, L. E. A. M., Cavalheiro, E. A., Tan, A. M. et al. (1993). Circuit mechanisms of seizures in the pilocarpine model of chronic epilepsy: Cell loss and mossy fiber sprouting. Epilepsia 34, 985-995. doi:10.1111/j.1528-1157.1993.tb02123.x
Millan, M. H., Chapman, A. G. and Meldrum, B. S. (1993). Extracellular amino acid levels in hippocampus during pilocarpine-induced seizures. Epilepsy Res 14, 139-148. doi:10.1016/0920-1211(93)90018-3
Miller, A. R., Hawkins, N. A., Mccollom, C. E. et al. (2014). Mapping genetic modifiers of survival in a mouse model of Dravet syndrome. Genes Brain Behav 13, 163-172. doi:10.1111/gbb.12099
Mohanraj, R. and Brodie, M. J. (2006). Diagnosing refractory epilepsy: Response to sequential treatment schedules. Eur J Neurol 13, 277-282. doi:10.1111/j.1468-1331.2006.01215.x
Mouri, G., Jimenez-Mateos, E., Engel, T. et al. (2008). Unilateral hippocampal CA3-predominant damage and short latency epileptogenesis after intra-amygdala microinjection of kainic acid in mice. Brain Res 1213, 140-151. doi:10.1016/j.brainres.2008.03.061
Niu, W. and Parent, J. M. (2020). Modeling genetic epilepsies in a dish. Dev Dyn 249, 56-75. doi:10.1002/dvdy.79
Oakley, J. C., Kalume, F., Yu, F. H. et al. (2009). Temperature- and age-dependent seizures in a mouse model of severe myoclonic epilepsy in infancy. Proc Natl Acad Sci 106, 3994-3999. doi:10.1073/pnas.0813330106
Ogiwara, I., Miyamoto, H., Morita, N. et al. (2007). Nav1.1 localizes to axons of parvalbumin-positive inhibitory interneurons: A circuit basis for epileptic seizures in mice carrying an Scn1a gene mutation. J Neurosci 27, 5903-5914. doi:10.1523/JNEUROSCI.5270-06.2007
Okanishi, T., Akiyama, T., Tanaka, S.-I. et al. (2014). Interictal high frequency oscillations correlating with seizure outcome in patients with widespread epileptic networks in tuberous sclerosis complex. Epilepsia 55, 1602-1610. doi:10.1111/epi.12761
Putnam, T. J. and Merritt, H. H. (1937). Experimental determination of the anticonvulsant properties of some phenyl derivatives. Science 85, 525-526. doi:10.1126/science.85.2213.525
Quadrato, G., Nguyen, T., Macosko, E. Z. et al. (2017). Cell diversity and network dynamics in photosensitive human brain organoids. Nature 545, 48-53. doi:10.1038/nature22047
Quraishi, I. H., Stern, S., Mangan, K. P. et al. (2019). An epilepsy-associated KCNT1 mutation enhances excitability of human iPSC-derived neurons by increasing slack KNa currents. J Neurosci 39, 7438-7449. doi:10.1523/JNEUROSCI.1628-18.2019
Remy, S., Gabriel, S., Urban, B. W. et al. (2003). A novel mechanism underlying drug resistance in chronic epilepsy. Ann Neurol 53, 469-479. doi:10.1002/ana.10473
Roopun, A. K., Simonotto, J. D., Pierce, M. L. et al. (2010). A nonsynaptic mechanism underlying interictal discharges in human epileptic neocortex. Proc Natl Acad Sci 107, 338-343. doi:10.1073/pnas.0912652107
Salmi, M., Bolbos, R., Bauer, S. et al. (2018). Transient microstructural brain anomalies and epileptiform discharges in mice defective for epilepsy and language-related NMDA receptor subunit gene Grin2a. Epilepsia 59, 1919-1930. doi:10.1111/epi.14543
Sandow, N., Kim, S., Raue, C. et al. (2015). Drug resistance in cortical and hippocampal slices from resected tissue of epilepsy patients: No significant impact of P-glycoprotein and multidrug resistance-associated proteins. Front Neurol 6, 30. doi:10.3389/fneur.2015.00030
Schmidt, D. and Löscher, W. (2005). Drug resistance in epilepsy: Putative neurobiologic and clinical mechanisms. Epilepsia 46, 858-877. doi:10.1111/j.1528-1167.2005.54904.x
Sharma, A. K., Jordan, W. H., Reams, R. Y. et al. (2008). Temporal profile of clinical signs and histopathologic changes in an F-344 rat model of kainic acid-induced mesial temporal lobe epilepsy. Toxicol Pathol 36, 932-943. doi:10.1177/0192623308326093
Shibley, H. and Smith, B. N. (2002). Pilocarpine-induced status epilepticus results in mossy fiber sprouting and spontaneous seizures in C57BL/6 and CD-1 mice. Epilepsy Res 49, 109-120. doi:10.1016/S0920-1211(02)00012-8
Simkin, D. and Kiskinis, E. (2018). Modeling pediatric epilepsy through iPSC-Based technologies. Epilepsy Curr 18, 240-245. doi:10.5698/1535-7597.18.4.240
Simon, A., Traub, R. D., Vladimirov, N. et al. (2014). Gap junction networks can generate both ripple-like and fast ripple-like oscillations. Eur J Neurosci 39, 46-60. doi:10.1111/ejn.12386
Sloviter, R. S. (2005). The neurobiology of temporal lobe epilepsy: Too much information, not enough knowledge. C R Biol 328, 143-153. doi:10.1016/j.crvi.2004.10.010
Sloviter, R. S. and Bumanglag, A. V. (2013). Defining “epileptogenesis” and identifying “antiepileptogenic targets” in animal models of acquired temporal lobe epilepsy is not as simple as it might seem. Neuropharmacology 69, 3-15. doi:10.1016/j.neuropharm.2012.01.022
Smart, S. L., Lopantsev, V., Zhang, C. L. et al. (1998). Deletion of the K(V)1.1 potassium channel causes epilepsy in mice. Neuron 20, 809-819. doi:10.1016/s0896-6273(00)81018-1
Staba, R. J. (2013). Pathological oscillations in the pharmacoresistant epileptic brain. In Pharmacoresistance In Epilepsy (27-46). New York, NY, USA: Springer New York. doi:10.1007/978-1-4614-6464-8_3
Sterlini, B., Fruscione, F., Baldassari, S. et al. (2020). Progress of induced pluripotent stem cell technologies to understand genetic epilepsy. Int J Mol Sci 21, 482. doi:10.3390/ijms21020482
Sun, Y., Paşca, S. P., Portmann, T. et al. (2016). A deleterious Nav1.1 mutation selectively impairs telencephalic inhibitory neurons derived from Dravet Syndrome patients. Elife 5, 1-26. doi:10.7554/eLife.13073
Sun, Y. and Dolmetsch, R. E. (2018). Investigating the therapeutic mechanism of cannabidiol in a human induced pluripotent stem cell (iPSC)-based model of Dravet syndrome. Cold Spring Harb Symp Quant Biol 83, 185-191. doi:10.1101/sqb.2018.83.038174
Sutula, T., Xiao-Xian, H., Cavazos, J. et al. (1988). Synaptic reorganization in the hippocampus induced by abnormal functional activity. Science 239, 1147-1150. doi:10.1126/science.2449733
Tanouye, M. A., Ferrus, A. and Fujita, S. C. (1981). Abnormal action potentials associated with the Shaker complex locus of Drosophila. Proc Natl Acad Sci 78, 6548-6552. doi:10.1073/pnas.78.10.6548
Toyoda, I., Bower, M. R., Leyva, F. et al. (2013). Early activation of ventral hippocampus and subiculum during spontaneous seizures in a rat model of temporal lobe epilepsy. J Neurosci 33, 11100-11115. doi:10.1523/jneurosci.0472-13.2013
Traub, R. D., Cunningham, M. O. and Whittington, M. A. (2011). Chemical synaptic and gap junctional interactions between principal neurons: Partners in epileptogenesis. Neural Networks 24, 515-525. doi:10.1016/j.neunet.2010.11.007
Traub, R. D., Cunningham, M. O. and Whittington, M. A. (2014). What is a seizure network? Very fast oscillations at the interface between normal and epileptic brain. In H. Scharfman and P. Buckmaster (eds.), Issues in Clinical Epileptology: A View from the Bench. Advances in Experimental Medicine and Biology (71-80). Volume 813. Dordrecht, The Netherlands: Springer. doi:10.1007/978-94-017-8914-1_6
Tsai, M.-H., Chuang, Y.-C., Chang, H.-W. et al. (2009). Factors predictive of outcome in patients with de novo status epilepticus. QJM 102, 57-62. doi:10.1093/qjmed/hcn149
Turski, W. A., Cavalheiro, E. A., Schwarz, M. et al. (1983). Limbic seizures produced by pilocarpine in rats: Behavioural, electroencephalographic and neuropathological study. Behav Brain Res 9, 315-335. doi:10.1016/0166-4328(83)90136-5
UKHO (2019). Annual Statistics of Scientific Procedures on Living Animals Great Britain 2018.
Upton, N., Blackburn, T. P., Campbell, C. A. et al. (1997). Profile of SB-204269, a mechanistically novel anticonvulsant drug, in rat models of focal and generalized epileptic seizures. Br J Pharmacol 121, 1679-1686. doi:10.1038/sj.bjp.0701330
van der Staay, F. J. (2006). Animal models of behavioral dysfunctions: Basic concepts and classifications, and an evaluation strategy. Brain Res Rev 52, 131-159. doi:10.1016/j.brainresrev.2006.01.006
van Gassen, K. L. I., Hessel, E. V. S., Ramakers, G. M. J. et al. (2008). Characterization of febrile seizures and febrile seizure susceptibility in mouse inbred strains. Genes Brain Behav 7, 578-586. doi:10.1111/j.1601-183X.2008.00393.x
VanLandingham, K. E., Heinz, E. R., Cavazos, J. E. et al. (1998). Magnetic resonance imaging evidence of hippocampal injury after prolonged focal febrile convulsions. Ann Neurol 43, 413-426. doi:10.1002/ana.410430403
Wang, J., Lin, Z. J., Liu, L. et al. (2017). Epilepsy-associated genes. Seizure 44, 11-20. doi:10.1016/j.seizure.2016.11.030
Wang, L., Wang, X., Yuan, J. et al. (2009). Nestin in the temporal neocortex of the intractable epilepsy patients. Neurochem Res 34, 574-580. doi:10.1007/s11064-008-9824-4
Wickham, J., Brödjegård, N. G., Vighagen, R. et al. (2018). Prolonged life of human acute hippocampal slices from temporal lobe epilepsy surgery. Sci Rep 8, 1-13. doi:10.1038/s41598-018-22554-9
Wiebe, S., Blume, W. T., Girvin, J. P. et al. (2001). A randomized, controlled trial of surgery for temporal-lobe epilepsy. N Engl J Med 345, 311-318. doi:10.1056/NEJM200108023450501
Wu, J. Y., Sankar, R., Lerner, J. T. et al. (2010). Removing interictal fast ripples on electrocorticography linked with seizure freedom in children. Neurology 75, 1686-1694. doi:10.1212/WNL.0b013e3181fc27d0
Yu, F. H., Mantegazza, M., Westenbroek, R. E. et al. (2006). Reduced sodium current in GABAergic interneurons in a mouse model of severe myoclonic epilepsy in infancy. Nat Neurosci 9, 1142-1149. doi:10.1038/nn1754
Zhang, C.-L., Messing, A. and Chiu, S. Y. (1999). Specific alteration of spontaneous GABAergic inhibition in cerebellar Purkinje cells in mice lacking the potassium channel Kv1.1. J Neurosci 19, 2852-2864. doi:10.1523/jneurosci.19-08-02852.1999
Zuberi, S. M., Eunson, L. H., Spauschus, A. et al. (1999). A novel mutation in the human voltage-gated potassium channel gene (Kv1.1) associates with episodic ataxia type 1 and sometimes with partial epilepsy. Brain 122, 817-825. doi:10.1093/brain/122.5.817