Modular micro-physiological human tumor/tissue models based on decellularized tissue for improved preclinical testing
Main Article Content
Abstract
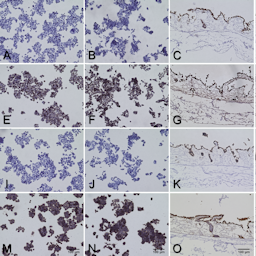
High attrition rates associated with drug testing in 2D cell culture and animal models stress the need for improved modeling of human tumor tissues. In previous studies, our 3D models on a decellularized tissue matrix have shown better predictivity and higher chemoresistance. A single porcine intestine yields material for 150 3D models of breast, lung, colorectal cancer (CRC) or leukemia. The uniquely preserved structure of the basement membrane enables physiological anchorage of endothelial cells and epithelial-derived carcinoma cells. The matrix provides different niches for cell growth: on top as monolayer, in crypts as aggregates, and within deeper layers. Dynamic culture in bioreactors enhances cell growth. Comparing gene expression between 2D and 3D cultures, we observed changes related to proliferation, apoptosis and stemness. For drug target predictions, we utilize tumor-specific sequencing data in our in silico model, finding an additive effect of metformin and gefitinib treatment for lung cancer in silico, validated in vitro. To analyze mode-of-action, immune therapies such as trispecific T-cell engagers in leukemia or toxicity on non-cancer cells, the model can be modularly enriched with human endothelial cells (hECs), immune cells and fibroblasts. Upon addition of hECs, transmigration of immune cells through the endothelial barrier can be investigated. In an allogenic CRC model, we observe a lower basic apoptosis rate after applying PBMCs in 3D compared to 2D, which offers new options to mirror antigen-specific immunotherapies in vitro. In conclusion, we present modular human 3D tumor models with tissue-like features for preclinical testing to reduce animal experiments.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Albritton, J. L. and Miller, J. S. (2017). 3D bioprinting: Improving in vitro models of metastasis with heterogeneous tumor microenvironments. Dis Model Mech 10, 3-14. doi:10.1242/dmm.025049
Anselmi, G., Vaivode, K., Dutertre, C. A. et al. (2020). Engineered niches support the development of human dendritic cells in humanized mice. Nat Commun 11, 2054. doi:10.1038/s41467-020-15937-y
Arrowsmith, J. and Miller, P. (2013). Trial watch: Phase II and phase III attrition rates 2011-2012. Nat Rev Drug Discov 12, 569. doi:10.1038/nrd4090
Baker, L. A., Tiriac, H., Clevers, H. et al. (2016). Modeling pancreatic cancer with organoids. Trends Cancer 2, 176-190. doi:10.1016/j.trecan.2016.03.004
Banaszek, A., Bumm, T. G. P., Nowotny, B. et al. (2019). On-target restoration of a split T cell-engaging antibody for precision immunotherapy. Nat Commun 10, 5387. doi:10.1038/s41467-019-13196-0
Baur, F., Nietzer, S. L., Kunz, M. et al. (2019). Connecting cancer pathways to tumor engines: A stratification tool for colorectal cancer combining human in vitro tissue models with Boolean in silico models. Cancers (Basel) 12, doi:10.3390/cancers12010028
Berger, C., Bjorlykke, Y., Hahn, L. et al. (2020). Matrix decoded – A pancreatic extracellular matrix with organ specific cues guiding human iPSC differentiation. Biomaterials 244, 119766. doi:10.1016/j.biomaterials.2020.119766
Bersi, H. (2017). Etablierung eines 3D in vitro Blutgefäß-/ Gewebemodells zur Testung spezifischer Therapeutika zur Leukämiebehandlung (Establishment of a 3D in vitro blood vessel / tissue model to test specific therapeutic agents to treat leukemia). University of Wuerzburg, medical faculty, urn:nbn:de:bvb:20-opus-152506.
Bhattacharjee, Y. (2012). Biomedicine. Pharma firms push for sharing of cancer trial data. Science 338, 29. doi:10.1126/science.338.6103.29
Bissell, M. J. (1981). The differentiated state of normal and malignant cells or how to define a “normal” cell in culture. Int Rev Cytol 70, 27-100. doi:10.1016/s0074-7696(08)61130-4
Bissell, M. J., Rizki, A. and Mian, I. S. (2003). Tissue architecture: The ultimate regulator of breast epithelial function. Curr Opin Cell Biol 15, 753-762. doi:10.1016/j.ceb.2003.10.016
Chae, Y. K., Arya, A., Malecek, M. K. et al. (2016). Repurposing metformin for cancer treatment: Current clinical studies. Oncotarget 7, 40767-40780. doi:10.18632/oncotarget.8194
Cree, I. A., Glaysher, S. and Harvey, A. L. (2010). Efficacy of anti-cancer agents in cell lines versus human primary tumour tissue. Curr Opin Pharmacol 10, 375-379. doi:10.1016/j.coph.2010.05.001
Di Cara, A., Garg, A., De Micheli, G. et al. (2007). Dynamic simulation of regulatory networks using squad. BMC Bioinformatics 8, 462. doi:10.1186/1471-2105-8-462
Dijkstra, K. K., Cattaneo, C. M., Weeber, F. et al. (2018). Generation of tumor-reactive T cells by co-culture of peripheral blood lymphocytes and tumor organoids. Cell 174, 1586-1598.e12. doi:10.1016/j.cell.2018.07.009
Drost, J. and Clevers, H. (2018). Organoids in cancer research. Nat Rev Cancer 18, 407-418. doi:10.1038/s41568-018-0007-6
EC European Commission (2020). 2019 report on the statistics on the use of animals for scientific purposes in the Member States of the European Union in 2015-2017. Brussels, 5.2.2020
Fan, H., Demirci, U. and Chen, P. (2019). Emerging organoid models: Leaping forward in cancer research. J Hematol Oncol 12, 142. doi:10.1186/s13045-019-0832-4
Francone, T. D., Landmann, R. G., Chen, C. T. et al. (2007). Novel xenograft model expressing human hepatocyte growth factor shows ligand-dependent growth of c-Met-expressing tumors. Mol Cancer Ther 6, 1460-1466. doi:10.1158/1535-7163.MCT-06-0466
Fuchs, M., Kreutzer, F. P., Kapsner, L. A. et al. (2020). Integrative bioinformatic analyses of global transcriptome data decipher novel molecular insights into cardiac anti-fibrotic therapies. Int J Mol Sci 21, 4727. doi:10.3390/ijms21134727
Fukuhara, S., Sako, K., Noda, K. et al. (2009). Tie2 is tied at the cell-cell contacts and to extracellular matrix by angiopoietin-1. Exp Mol Med 41, 133-139. doi:10.3858/emm.2009.41.3.016
Galon, J., Costes, A., Sanchez-Cabo, F. et al. (2006). Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313, 1960-1964. doi:10.1126/science.1129139
Gao, Q., Yang, Z., Xu, S. et al. (2019). Heterotypic CAF-tumor spheroids promote early peritoneal metastatis of ovarian cancer. J Exp Med 216, 688-703. doi:10.1084/jem.20180765
Goebeler, M. E. and Bargou, R. C. (2020). T cell-engaging therapies – Bites and beyond. Nat Rev Clin Oncol 17, 418-434. doi:10.1038/s41571-020-0347-5
Göttlich, C., Muller, L. C., Kunz, M. et al. (2016). A combined 3D tissue engineered in vitro/in silico lung tumor model for predicting drug effectiveness in specific mutational backgrounds. J Vis Exp, e53885. doi:10.3791/53885
Göttlich, C., Kunz, M., Zapp, C. et al. (2018). A combined tissue-engineered/in silico signature tool patient stratification in lung cancer. Mol Oncol 12, 1264-1285. doi:10.1002/1878-0261.12323
Groeber, F., Engelhardt, L., Lange, J. et al. (2016). A first vascularized skin equivalent as an alternative to animal experimentation. ALTEX 33, 415-422. doi:10.14573/altex.1604041
Gubbiotti, M. A., Buraschi, S., Kapoor, A. et al. (2020). Proteoglycan signaling in tumor angiogenesis and endothelial cell autophagy. Semin Cancer Biol 62, 1-8. doi:10.1016/j.semcancer.2019.05.003
Howat, W. J., Barabas, T., Holmes, J. A. et al. (2002). Distribution of basement membrane pores in bronchus revealed by microscopy following epithelial removal. J Struct Biol 139, 137-145. doi:10.1016/s1047-8477(02)00589-0
Inwald, E. C., Klinkhammer-Schalke, M., Hofstadter, F. et al. (2013). Ki-67 is a prognostic parameter in breast cancer patients: Results of a large population-based cohort of a cancer registry. Breast Cancer Res Treat 139, 539-552. doi:10.1007/s10549-013-2560-8
Jannasch, M., Groeber, F., Brattig, N. W. et al. (2015). Development and application of three-dimensional skin equivalents for the investigation of percutaneous worm invasion. Exp Parasitol 150, 22-30. doi:10.1016/j.exppara.2015.01.005
Jeffers, M., Rong, S. and Vande Woude, G. F. (1996). Hepatocyte growth factor/scatter factor-met signaling in tumorigenicity and invasion/metastasis. J Mol Med (Berl) 74, 505-513. doi:10.1007/BF00204976
Kalluri, R. (2016). The biology and function of fibroblasts in cancer. Nat Rev Cancer 16, 582-598. doi:10.1038/nrc.2016.73
Katt, M. E., Placone, A. L., Wong, A. D. et al. (2016). In vitro tumor models: Advantages, disadvantages, variables, and selecting the right platform. Front Bioeng Biotechnol 4, 12. doi:10.3389/fbioe.2016.00012
Kim, M., Mun, H., Sung, C. O. et al. (2019). Patient-derived lung cancer organoids as in vitro cancer models for therapeutic screening. Nat Commun 10, 3991. doi:10.1038/s41467-019-11867-6
Levin, T. G., Powell, A. E., Davies, P. S. et al. (2010). Characterization of the intestinal cancer stem cell marker CD166 in the human and mouse gastrointestinal tract. Gastroenterology 139, 2072-2082.e5. doi:10.1053/j.gastro.2010.08.053
Linke, K., Schanz, J., Hansmann, J. et al. (2007). Engineered liver-like tissue on a capillarized matrix for applied research. Tissue Eng 13, 2699-2707. doi:10.1089/ten.2006.0388
Liu, D., Sun, J., Zhu, J. et al. (2014). Expression and clinical significance of colorectal cancer stem cell marker EpCAMhigh/CD44+ in colorectal cancer. Oncol Lett 7, 1544-1548. doi:10.3892/ol.2014.1907
Lu, S., Wang, H. and Taube, J. M. (2019). Different biomarker modalities and response to anti-PD-1/PD-L1 therapies-reply. JAMA Oncol doi:10.1001/jamaoncol.2019.5154
Matsuoka, Y., Funahashi, A., Ghosh, S. et al. (2014). Modeling and simulation using celldesigner. Methods Mol Biol 1164, 121-145. doi:10.1007/978-1-4939-0805-9_11
McClugage, S. G. and Low, F. N. (1984). Microdissection by ultrasonication: Porosity of the intestinal epithelial basal lamina. Am J Anat 171, 207-216. doi:10.1002/aja.1001710206
Meurette, O. and Mehlen, P. (2018). Notch signaling in the tumor microenvironment. Cancer Cell 34, 536-548. doi:10.1016/j.ccell.2018.07.009
Mullins, C. S., Micheel, B., Matschos, S. et al. (2019). Integrated biobanking and tumor model establishment of human colorectal carcinoma provides excellent tools for preclinical research. Cancers (Basel) 11, 1520. doi:10.3390/cancers11101520
Munz, M., Baeuerle, P. A. and Gires, O. (2009). The emerging role of EpCAM in cancer and stem cell signaling. Cancer Res 69, 5627-5629. doi:10.1158/0008-5472.CAN-09-0654
Nath, S. and Devi, G. R. (2016). Three-dimensional culture systems in cancer research: Focus on tumor spheroid model. Pharmacol Ther 163, 94-108. doi:10.1016/j.pharmthera.2016.03.013
Neve, R. M., Chin, K., Fridlyand, J. et al. (2006). A collection of breast cancer cell lines for the study of functionally distinct cancer subtypes. Cancer Cell 10, 515-527. doi:10.1016/j.ccr.2006.10.008
Nietzer, S., Baur, F., Sieber, S. et al. (2016). Mimicking metastases including tumor stroma: A new technique to generate a three-dimensional colorectal cancer model based on a biological decellularized intestinal scaffold. Tissue Eng Part C Methods 22, 621-635. doi:10.1089/ten.TEC.2015.0557
Nusgens, B. V. (2010). Hyaluronic acid and extracellular matrix: A primitive molecule? [Article in French]. Ann Dermatol Venereol 137, Suppl 1, S3-8. doi:10.1016/S0151-9638(10)70002-8
Raudvere, U., Kolberg, L., Kuzmin, I. et al. (2019). g:Profiler: A web server for functional enrichment analysis and conversions of gene lists (2019 update). Nucleic Acids Res 47, W191-W198. doi:10.1093/nar/gkz369
Rongvaux, A., Takizawa, H., Strowig, T. et al. (2013). Human hemato-lymphoid system mice: Current use and future potential for medicine. Annu Rev Immunol 31, 635-674. doi:10.1146/annurev-immunol-032712-095921
Rongvaux, A., Willinger, T., Martinek, J. et al. (2014). Development and function of human innate immune cells in a humanized mouse model. Nat Biotechnol 32, 364-372. doi:10.1038/nbt.2858
Sahlberg, S. H., Spiegelberg, D., Glimelius, B. et al. (2014). Evaluation of cancer stem cell markers CD133, CD44, CD24: Association with AKT isoforms and radiation resistance in colon cancer cells. PLoS One 9, e94621. doi:10.1371/journal.pone.0094621
Santo, V. E., Rebelo, S. P., Estrada, M. F. et al. (2017). Drug screening in 3D in vitro tumor models: Overcoming current pitfalls of efficacy read-outs. Biotechnol J 12. doi:10.1002/biot.201600505
Schanz, J., Pusch, J., Hansmann, J. et al. (2010). Vascularised human tissue models: A new approach for the refinement of biomedical research. J Biotechnol 148, 56-63. doi:10.1016/j.jbiotec.2010.03.015
Schweinlin, M., Rossi, A., Lodes, N. et al. (2017). Human barrier models for the in vitro assessment of drug delivery. Drug Deliv Transl Res 7, 217-227. doi:10.1007/s13346-016-0316-9
Shibue, T. and Weinberg, R. A. (2017). EMT, CSCs, and drug resistance: The mechanistic link and clinical implications. Nat Rev Clin Oncol 14, 611-629. doi:10.1038/nrclinonc.2017.44
Simian, M. and Bissell, M. J. (2017). Organoids: A historical perspective of thinking in three dimensions. J Cell Biol 216, 31-40. doi:10.1083/jcb.201610056
Socinski, M. A., Goldman, J., El-Hariry, I. et al. (2013). A multicenter phase II study of ganetespib monotherapy in patients with genotypically defined advanced non-small cell lung cancer. Clin Cancer Res 19, 3068-3077. doi:10.1158/1078-0432.CCR-12-3381
Song, Y., Rongvaux, A., Taylor, A. et al. (2019). A highly efficient and faithful MDS patient-derived xenotransplantation model for pre-clinical studies. Nat Commun 10, 366. doi:10.1038/s41467-018-08166-x
Sos, M. L., Michel, K., Zander, T. et al. (2009). Predicting drug susceptibility of non-small cell lung cancers based on genetic lesions. J Clin Invest 119, 1727-1740. doi:10.1172/JCI37127
Stieglitz, E., Ward, A. F., Gerbing, R. B. et al. (2015). Phase II/III trial of a pre-transplant farnesyl transferase inhibitor in juvenile myelomonocytic leukemia: A report from the children’s oncology group. Pediatr Blood Cancer 62, 629-636. doi:10.1002/pbc.25342
Stratmann, A. T., Fecher, D., Wangorsch, G. et al. (2014). Establishment of a human 3D lung cancer model based on a biological tissue matrix combined with a Boolean in silico model. Mol Oncol 8, 351-365. doi:10.1016/j.molonc.2013.11.009
Stuber, T., Monjezi, R., Wallstabe, L. et al. (2020). Inhibition of TGF-beta-receptor signaling augments the antitumor function of ROR1-specific CAR T-cells against triple-negative breast cancer. J Immunother Cancer 8, e000676. doi:10.1136/jitc-2020-000676
Sun, J., Zhao, M., Jia, P. et al. (2015). Deciphering signaling pathway networks to understand the molecular mechanisms of metformin action. PLoS Comput Biol 11, e1004202. doi:10.1371/journal.pcbi.1004202
Takahashi-Iwanaga, H., Iwanaga, T. and Isayama, H. (1999). Porosity of the epithelial basement membrane as an indicator of macrophage-enterocyte interaction in the intestinal mucosa. Arch Histol Cytol 62, 471-481. doi:10.1679/aohc.62.471
Takeuchi, T. and Gonda, T. (2004). Distribution of the pores of epithelial basement membrane in the rat small intestine. J Vet Med Sci 66, 695-700. doi:10.1292/jvms.66.695
Thomas, X. and Elhamri, M. (2007). Tipifarnib in the treatment of acute myeloid leukemia. Biologics 1, 415-424.
Tumeh, P. C., Harview, C. L., Yearley, J. H. et al. (2014). PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515, 568-571. doi:10.1038/nature13954
Valkenburg, K. C., de Groot, A. E. and Pienta, K. J. (2018). Targeting the tumour stroma to improve cancer therapy. Nat Rev Clin Oncol 15, 366-381. doi:10.1038/s41571-018-0007-1
van Duinen, V., Trietsch, S. J., Joore, J. et al. (2015). Microfluidic 3D cell culture: From tools to tissue models. Curr Opin Biotechnol 35, 118-126. doi:10.1016/j.copbio.2015.05.002
Wallstabe, L., Göttlich, C., Nelke, L. C. et al. (2019). ROR1-car T cells are effective against lung and breast cancer in advanced microphysiologic 3D tumor models. JCI Insight 4, e126345. doi:10.1172/jci.insight.126345
Walsh, N. C., Kenney, L. L., Jangalwe, S. et al. (2017). Humanized mouse models of clinical disease. Annu Rev Pathol 12, 187-215. doi:10.1146/annurev-pathol-052016-100332
Wang, C., Xie, J., Guo, J. et al. (2012). Evaluation of CD44 and CD133 as cancer stem cell markers for colorectal cancer. Oncol Rep 28, 1301-1308. doi:10.3892/or.2012.1951
Wang, L., Zuo, X., Xie, K. et al. (2018). The role of CD44 and cancer stem cells. Methods Mol Biol 1692, 31-42. doi:10.1007/978-1-4939-7401-6_3
Warth, A., Cortis, J., Soltermann, A. et al. (2014). Tumour cell proliferation (Ki-67) in non-small cell lung cancer: A critical reappraisal of its prognostic role. Br J Cancer 111, 1222-1229. doi:10.1038/bjc.2014.402
Weeber, F., Ooft, S. N., Dijkstra, K. K. et al. (2017). Tumor organoids as a pre-clinical cancer model for drug discovery. Cell Chem Biol 24, 1092-1100. doi:10.1016/j.chembiol.2017.06.012
Wickham, H. (2009). ggplot2 – Elegant Graphics for Data Analysis. Germany: Springer.
Willis, N. D., Przyborski, S. A., Hutchison, C. J. et al. (2008). Colonic and colorectal cancer stem cells: Progress in the search for putative biomarkers. J Anat 213, 59-65. doi:10.1111/j.1469-7580.2008.00917.x
Xu, X., Farach-Carson, M. C. and Jia, X. (2014). Three-dimensional in vitro tumor models for cancer research and drug evaluation. Biotechnol Adv 32, 1256-1268. doi:10.1016/j.biotechadv.2014.07.009
Yada, E., Wada, S., Yoshida, S. et al. (2018). Use of patient-derived xenograft mouse models in cancer research and treatment. Future Sci OA 4, FSO271. doi:10.4155/fsoa-2017-0136
Zhang, Y. and Weinberg, R. A. (2018). Epithelial-to-mesenchymal transition in cancer: Complexity and opportunities. Front Med 12, 361-373. doi:10.1007/s11684-018-0656-6