A new approach methodology (NAM) for the prediction of (nor)ibogaine-induced cardiotoxicity in humans
Main Article Content
Abstract
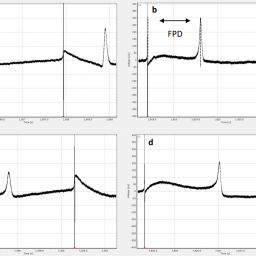
The development of non-animal-based new approach methodologies (NAMs) for chemical risk assessment and safety evaluation is urgently needed. The aim of the present study was to investigate the applicability of an in vitro-in silico approach to predict human cardiotoxicity of the herbal alkaloid ibogaine and its metabolite noribogaine, which are promising anti-addiction drugs. Physiologically based kinetic (PBK) models were developed using in silico-derived parameters and biokinetic data obtained from in vitro liver microsomal incubations and Caco-2 transport studies. Human induced pluripotent stem cell-derived cardiomyocytes combined with a multi-electrode array (MEA) assay were used to determine in vitro concentration-dependent cardiotoxicity reflected by prolongation of field potential duration, which was subsequently translated to in vivo dose-dependent prolongation of the QTc (heart rate corrected duration from ventricular depolarization to repolarization) using PBK modeling-based reverse dosimetry. Results showed that the predictions matched well with in vivo kinetic data and QTc data for ibogaine and noribogaine available in the literature, indicating a good performance of the NAM. Benchmark dose analysis of the predicted dose response curves adequately predicted the onset of in vivo cardiotoxicity detected by QTc prolongation upon oral exposure to ibogaine and noribogaine. The present study provides an additional proof-of-principle of using PBK modeling-based reverse dosimetry as a NAM to predict human cardiotoxicity.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Abdullah, R., Alhusainy, W., Woutersen, J. et al. (2016) Predicting points of departure for risk assessment based on in vitro cytotoxicity data and physiologically based kinetic (PBK) modeling: the case of kidney toxicity induced by aristolochic acid I. Food and chemical toxicology 92:104-116. doi:10.1016/j.fct.2016.03.017
Alper, K.R., Bai, R., Liu, N. et al. (2016) hERG blockade by iboga alkaloids. Cardiovascular toxicology 16(1):14-22. doi:10.1007/s12012-015-9311-5
Alper, K.R., Lotsof, H. S., Geerte, M. N. et al. (1999) Treatment of acute opioid withdrawal with ibogaine. American Journal on Addictions 8(3):234-242. doi:10.1080/105504999305848
Alper, K.R., Stajić, M., Gill, J.R. (2012) Fatalities temporally associated with the ingestion of ibogaine. Journal of forensic sciences 57(2):398-412. doi:10.1111/j.1556-4029.2011.02008.x
Andersen, M. E., McMullen, P.D., Phillips, M. B. et al. (2019) Developing context appropriate toxicity testing approaches using new alternative methods (NAMs). ALTEX-Alternatives to animal experimentation 36(4):523-534. doi:10.14573/altex.1906261
Ando, H., Yoshinaga, T., Yamamoto, W. et al. (2017) A new paradigm for drug-induced torsadogenic risk assessment using human iPS cell-derived cardiomyocytes. Journal of pharmacological and toxicological methods 84:111-127. doi:10.1016/j.vascn.2016.12.003
Asua, I. (2013) Growing menace of ibogaine toxicity. British journal of anaesthesia 111(6):1029-1030. doi:10.1093/bja/aet396
Badhan RK, Gittins R, Al Zabit D (2019) The optimization of methadone dosing whilst treating with rifampicin: a pharmacokinetic modeling study. Drug Alcohol Depend 200:168–180 doi:10.1016/j.drugalcdep.2019.03.013
Baumann, M. H., Pablo, J., Ali, S. F. et al. (2001a) Comparative neuropharmacology of ibogaine and its O-desmethyl metabolite, noribogaine. In K. Alper, G. A. Cordell (ed.) The Alkaloids Chemistry and biology 56 (79-113). Amsterdam, The Netherlands: Elsevier. doi:10.1016/S0099-9598(01)56009-5
Baumann, M. H., Rothman, R. B., Pablo, J. P., Mash, D. C. (2001b) In vivo neurobiological effects of ibogaine and its O-desmethyl metabolite, 12-hydroxyibogamine (noribogaine), in rats. Journal of Pharmacology and Experimental Therapeutics 297(2):531-539. https://pubmed.ncbi.nlm.nih.gov/11303040/
Bentz, J., O'Connor, M. P., Bednarczyk, D. et al. (2013) Variability in P-glycoprotein inhibitory potency (IC50) using various in vitro experimental systems: implications for universal digoxin drug-drug interaction risk assessment decision criteria. Drug Metabolism and Disposition 41(7):1347-1366. doi:10.1124/dmd.112.050500
Bos, P. M., Geraets, L., de Wit-Bos, L. et al. (2020) Towards an animal-free human health assessment: Starting from the current regulatory needs. ALTEX-Alternatives to animal experimentation 37(3):395-408. doi:10.14573/altex.1912041
Brown, R. P., Delp, M. D., Lindstedt, S. L. et al. (1997) Physiological parameter values for physiologically based pharmacokinetic models. Toxicology and industrial health 13(4):407-484. doi:10.1177/074823379701300401
Chang, Y., Fang, W. B., Lin, S. N., Moody, D. E. (2011). Stereo‐selective metabolism of methadone by human liver microsomes and cDNA‐expressed cytochrome P450s: a reconciliation. Basic & clinical pharmacology & toxicology 108(1): 55-62. doi:10.1111/j.1742-7843.2010.00628.x
Davis, A. K., Barsuglia, J. P., Windham-Herman, A. M. et al. (2017) Subjective effectiveness of ibogaine treatment for problematic opioid consumption: short-and long-term outcomes and current psychological functioning. Journal of psychedelic studies 1(2):65-73. doi:10.1556/2054.01.2017.009
EFSA – European Food Safety Authority (2017) Update: use of the benchmark dose approach in risk assessment. doi:10.2903/j.efsa.2017.4658
Fisher, M. B., Campanale, K., Ackermann, B. L. et al. (2000) In vitro glucuronidation using human liver microsomes and the pore-forming peptide alamethicin. Drug metabolism and disposition 28(5):560-566.
Florian, J., Garnett, C., Nallani, S. et al. (2012) A modeling and simulation approach to characterize methadone QT prolongation using pooled data from five clinical trials in MMT patients. Clinical Pharmacology & Therapeutics 91:666-672. doi:10.1038/clpt.2011.273
Foster, D. J., Somogyi, A. A., Dyer, K. R. et al. (2000) Steady‐state pharmacokinetics of (R)‐and (S)‐methadone in methadone maintenance patients. British journal of clinical pharmacology 50(5):427-440. doi:10.1046/j.1365-2125.2000.00272.x
Ferruzza S., Rossi C., Sambuy, Y., Scarino M. L. (2013). Serum-reduced and serum-free media for differentiation of Caco-2 cells. ALTEX-Alternatives to animal experimentation, 30(2), 159-168. doi:10.14573/altex.2013.2.159
Gilbert‐Sandoval, I., Wesseling, S., Rietjens, I. M. C. M. (2020) Predicting the Acute Liver Toxicity of Aflatoxin B1 in Rats and Humans by an In Vitro-In Silico Testing Strategy. Molecular nutrition & food research 64(13):2000063. doi:10.1002/mnfr.202000063
Glue, P., Cape, G., Tunnicliff, D. et al. (2016) Ascending single‐dose, double‐blind, placebo‐controlled safety study of noribogaine in opioid‐dependent patients. Clinical pharmacology in drug development 5(6):460-468. doi:10.1002/cpdd.254
Glue, P., Lockhart, M., Lam, F. et al. (2015a) Ascending‐dose study of noribogaine in healthy volunteers: Pharmacokinetics, pharmacodynamics, safety, and tolerability. The Journal of Clinical Pharmacology 55(2):189-194. doi:10.1002/jcph.404
Glue, P., Winter, H., Garbe, K. et al. (2015b) Influence of CYP2D6 activity on the pharmacokinetics and pharmacodynamics of a single 20 mg dose of ibogaine in healthy volunteers. The Journal of Clinical Pharmacology 55(6):680-687. doi:10.1002/jcph.471
Goutarel, R., Gollnhofer, O., Sillans, R. (1993) Pharmacodynamics and therapeutic applications of iboga and ibogaine. Psychedelic Monographs and Essays 6 (71-111) https://ibogainedossier.com/bwiti1.html
Grogan, J., Gerona, R., Snow, J. W., Kao, L. (2019) Ibogaine Consumption With Seizure-Like Episodes, QTc-Prolongation, and Captured Cardiac Dysrhythmias. The Journal of emergency medicine 57(4):e99-e104. doi:10.1016/j.jemermed.2019.06.05
Henstra, M., Wong, L., Chahbouni, A., Swart, N. et al. (2017) Toxicokinetics of ibogaine and noribogaine in a patient with prolonged multiple cardiac arrhythmias after ingestion of internet purchased ibogaine. Clinical Toxicology 55(6):600-602. doi:10.1080/15563650.2017.1287372
Hildyard, C., Macklin, P., Prendergast, B., Bashir, Y. (2016) A case of QT prolongation and torsades de pointes caused by ibogaine toxicity. Journal of Emergency Medicine 50(2):e83-e87. doi:10.1016/j.jemermed.2015.06.05
Hoelen, D. W., Spiering, W., Valk, G. D. (2009) Long-QT syndrome induced by the antiaddiction drug ibogaine. New England journal of medicine 360(3):308-309. doi:10.1056/NEJMc0804248
Hubatsch, I., Ragnarsson, E. G., Artursson, P. (2007) Determination of drug permeability and prediction of drug absorption in Caco-2 monolayers. Nature protocols 2(9):2111. doi:10.1038/nprot.2007.303
ICCVAM – Interagency Coordinating Committee on the Validation of Alternative Methods (2018). A Strategic Roadmap for Establishing New Approaches to Evaluate the Safety of Chemicals and Medical Products in the United States. doi:10.22427/NTP-ICCVAM-ROADMAP2018.
ICH – The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (2005) E14: the clinical evaluation of QT/QTc interval prolongation and proarrhythmic potential for non-antiarrhythmic drugs. https://database.ich.org/sites/default/files/E14_Guideline.pdf Accessed 20 Jan 2021
Karakikes, I., Ameen, M., Termglinchan, V., Wu, J. C. (2015) Human induced pluripotent stem cell-derived cardiomyocytes: insights into molecular, cellular, and functional phenotypes. Circulation research 117(1):80-88. doi:10.1161/CIRCRESAHA.117.305365
Kitaguchi, T., Moriyama, Y., Taniguchi, T. et al. (2017) CSAHi study: detection of drug-induced ion channel/receptor responses, QT prolongation, and arrhythmia using multi-electrode arrays in combination with human induced pluripotent stem cell-derived cardiomyocytes. Journal of pharmacological and toxicological methods 85:73-81. doi:10.1016/j.vascn.2017.02.001
Koenig, X., Kovar, M., Boehm, S. et al. (2014) Anti‐addiction drug ibogaine inhibits hERG channels: a cardiac arrhythmia risk. Addiction biology 19(2):237-239. doi:10.1111/j.1369-1600.2012.00447.x
Kraushaar U, Guenther E (2019) Assay procedures for compound testing of hiPSC-derived cardiomyocytes using multiwell microelectrode arrays Cell-Based Assays Using iPSCs for Drug Development and Testing. Springer, p 197-208. doi:10.1007/978-1-4939-9477-9_18
Kussauer, S., David, R., Lemcke, H. (2019) hiPSCs derived cardiac cells for drug and toxicity screening and disease modeling: what micro-electrode-array analyses can tell us. Cells 8(11):1331. doi:10.3390/cells8111331
Litjens, R. P., Brunt, T. M. (2016) How toxic is ibogaine? Clinical Toxicology 54(4):297-302. doi:10.3109/15563650.2016.1138226
Louisse, J., de Jong, E., van de Sandt, J. J. et al. (2010) The use of in vitro toxicity data and physiologically based kinetic modeling to predict dose-response curves for in vivo developmental toxicity of glycol ethers in rat and man. Toxicological Sciences 118(2):470-484. doi:10.1093/toxsci/kfq270
Ma, J., Guo, L., Fiene, S. J. et al. (2011) High purity human-induced pluripotent stem cell-derived cardiomyocytes: electrophysiological properties of action potentials and ionic currents. American Journal of Physiology-Heart and Circulatory Physiology 301(5):H2006-H2017. doi:10.1152/ajpheart.00694.2011
Maciulaitis, R., Kontrimaviciute, V., Bressolle, F., Briedis, V. (2008) Ibogaine, an anti-addictive drug: pharmacology and time to go further in development. A narrative review. Human and Experimental Toxicology 27(3):181. doi:10.1177/0960327107087802
Martin, R. L., McDermott, J. S., Salmen, H. J. et al. (2004) The utility of hERG and repolarization assays in evaluating delayed cardiac repolarization: influence of multi-channel block. Journal of cardiovascular pharmacology 43(3):369-379. doi:10.1097/00005344-200403000-00007
Mash, D. C., Kovera, C. A., Pablo, J. et al. (2001). Ibogaine in the treatment of heroin withdrawal. The Alkaloids Chemistry and biology 56 (155-171). Amsterdam, The Netherlands: Elsevier. doi:10.1016/S0099-9598(01)56012-5
Mash, D. C., Ameer, B., Prou, D. et al. (2016) Oral noribogaine shows high brain uptake and anti-withdrawal effects not associated with place preference in rodents. Journal of Psychopharmacology 30(7):688-697. doi:10.1177/0269881116641331
Mash, D. C., Duque, L., Page, B., Allen-Ferdinand. K. (2018) Ibogaine detoxification transitions opioid and cocaine abusers between dependence and abstinence: clinical observations and treatment outcomes. Frontiers in pharmacology 9:529. doi:10.3389/fphar.2018.00529
Meisner, J. A., Wilcox, S. R., Richards, J. B. (2016) Ibogaine-associated cardiac arrest and death: case report and review of the literature. Therapeutic advances in psychopharmacology 6(2):95-98. doi:10.1177/2045125315626073
Mujtaba, S., Romero, J., Taub, C. C. (2013) Methadone, QTc prolongation and torsades de pointes: current concepts, management and a hidden twist in the tale? Journal of Cardiovascular Disease Research 4:229-235. doi:10.1016/j.jcdr.2013.10.001
Ning, J., Louisse, J., Spenkelink, B. et al. (2017) Study on inter-ethnic human differences in bioactivation and detoxification of estragole using physiologically based kinetic modeling. Archives of toxicology 91(9):3093-3108. doi:10.1007/s00204-017-1941-x
Noller, G. E, Frampton, C. M., Yazar-Klosinski, B. (2018) Ibogaine treatment outcomes for opioid dependence from a twelve-month follow-up observational study. The American journal of drug and alcohol abuse 44(1):37-46. doi:10.1080/00952990.2017.1310218
Nozaki, Y., Honda, Y., Watanabe, H. et al. (2017) CSAHi study-2: validation of multi-electrode array systems (MEA60/2100) for prediction of drug-induced proarrhythmia using human iPS cell-derived cardiomyocytes: assessment of reference compounds and comparison with non-clinical studies and clinical information. Regulatory Toxicology and Pharmacology 88:238-251. doi:10.1016/j.yrtph.2017.06.006
O'Connell, C. W., Gerona, R. R, Friesen, M. W, Ly, B. T. (2015) Internet-purchased ibogaine toxicity confirmed with serum, urine, and product content levels. The American journal of emergency medicine 33(7):985. e5-985. e6. doi:10.1016/j.ajem.2014.12.023
Obach, R. S, Pablo, J., Mash, D. C. (1998) Cytochrome P4502D6 catalyzes the O-demethylation of the psychoactive alkaloid ibogaine to 12-hydroxyibogamine. Drug metabolism and disposition 26(8):764-768.
Ovics, P., Regev, D., Baskin, P. et al. (2020) Drug Development and the Use of Induced Pluripotent Stem Cell-Derived Cardiomyocytes for Disease Modeling and Drug Toxicity Screening. International Journal of Molecular Sciences 21(19):7320. doi:10.3390/ijms21197320
Paling, F., Andrews, L., Valk, G., Blom, H. (2012) Life-threatening complications of ibogaine: three case reports. The Netherlands Journal of Medicine 70(9):422-4.
Pang, L., Sager, P., Yang, X. et al. (2019) Workshop report: FDA workshop on improving cardiotoxicity assessment with human-relevant platforms. Circulation research 125(9):855-867. doi:10.1161/CIRCRESAHA.119.315378
Patterson, E. A., Whelan, M. P., Worth, A. P. (2020) The role of validation in establishing the scientific credibility of predictive toxicology approaches intended for regulatory application. Computational Toxicology:100144. doi:10.1016/j.comtox.2020.100144
Pleskovic, A., Gorjup, V., Brvar, M., Kozelj, G. (2012) Ibogaine-associated ventricular tachyarrhythmias. Clinical Toxicology 50 (2): 157-157. doi:10.3109/15563650.2011.647031
Punt, A., Bouwmeester, H., Blaauboer, B. J. et al. (2020) New approach methodologies (NAMs) for human-relevant biokinetics predictions: Meeting the paradigm shift in toxicology towards an animal-free chemical risk assessment. ALTEX-Alternatives to animal experimentation 37(4):607-622. doi:10.14573/altex.2003242
Rietjens, I. M. C. M., Louisse, J., Punt, A. (2011) Tutorial on physiologically based kinetic modeling in molecular nutrition and food research. Molecular nutrition & food research 55(6):941-956. doi:10.1002/mnfr.201000655
Rohatgi, A. (2020). WebPlotDigitizer (Version 4.4) [Computer software]. Retrieved from https://apps.automeris.io/wpd/
Rubi, L., Eckert, D., Boehm, S. et al. (2017) Anti-addiction drug ibogaine prolongs the action potential in human induced pluripotent stem cell-derived cardiomyocytes. Cardiovascular toxicology 17(2):215-218. doi:10.1007/s12012-016-9366-y
Sala, L., Ward-van Oostwaard, D., Tertoolen, L. G. et al. (2017) Electrophysiological analysis of human pluripotent stem cell-derived cardiomyocytes (hPSC-CMs) using multi-electrode arrays (MEAs). JoVE (Journal of Visualized Experiments)(123):e55587. doi:10.3791/55587
Satsuka, A., Kanda, Y. (2020) Cardiotoxicity Assessment of Drugs Using Human iPS Cell-Derived Cardiomyocytes: Toward Proarrhythmic Risk and Cardio-Oncology. Current pharmaceutical biotechnology 21(9):765-772. doi:10.2174/1389201020666190628143345
Schep, L. J., Slaughter, R., Galea, S., Newcombe, D. (2016) Ibogaine for treating drug dependence. What is a safe dose? Drug and alcohol dependence 166:1-5. doi:10.1016/j.drugalcdep.2016.07.005
Shi, M., Bouwmeester, H., Rietjens, I. M. C. M., Strikwold, M. (2020a) Integrating in vitro data and physiologically based kinetic modeling-facilitated reverse dosimetry to predict human cardiotoxicity of methadone. Archives of toxicology 94(8):2809-2827. doi:10.1007/s00204-020-02766-7
Shi, M., Tien, N. T., de Haan, L. et al. (2020b). Evaluation of in vitro models of stem cell-derived cardiomyocytes to screen for potential cardiotoxicity of chemicals. Toxicology in Vitro: 104891. doi:10.1016/j.tiv.2020.104891
Smith, D. A., Di, L., Kerns, E. H. (2010) The effect of plasma protein binding on in vivo efficacy: misconceptions in drug discovery. Nature reviews Drug discovery 9(12):929-939. doi:10.1038/nrd3287
Smith, S. A., Waters, N. J. (2019) Pharmacokinetic and pharmacodynamic considerations for drugs binding to alpha-1-acid glycoprotein. Pharmaceutical research 36(2):1-19. doi:10.1007/s11095-018-2551-x
Steinberg, C., Deyell, M. W. (2018) Cardiac arrest after ibogaine intoxication. Journal of arrhythmia 34(4):455-457. doi:10.1002/joa3.12061
Strikwold, M., Spenkelink, B., de Haan, L. et al. (2017) Integrating in vitro data and physiologically based kinetic (PBK) modelling to assess the in vivo potential developmental toxicity of a series of phenols. Archives of toxicology 91(5):2119-2133. doi:10.1007/s00204-016-1881-x
Taboureau, O., El M'Selmi, W., Audouze, K. (2020) Integrative systems toxicology to predict human biological systems affected by exposure to environmental chemicals. Toxicology and Applied Pharmacology 405:115210. doi:10.1016/j.taap.2020.115210
van Liempd, S., Morrison, D., Sysmans, L. et al. (2011) Development and validation of a higher-throughput equilibrium dialysis assay for plasma protein binding. JALA: Journal of the Association for Laboratory Automation 16(1):56-67. doi:10.1016/j.jala.2010.06.002
Vandenberk B, Vandael E, Robyns T, et al. (2016) Which QT correction formulae to use for QT monitoring? Journal of the American Heart Association 5(6):e003264. doi:10.1161/JAHA.116.003264
Vlaanderen, L., Martial, L., Franssen, E. et al. (2014) Cardiac arrest after ibogaine ingestion. Clinical Toxicology 52(6):642-643. doi:10.3109/15563650.2014.927477
Waters, N. J., Jones, R., Williams, G., Sohal, B. (2008) Validation of a rapid equilibrium dialysis approach for the measurement of plasma protein binding. Journal of pharmaceutical sciences 97(10):4586-4595. doi:10.1002/jps.21317
Wedam, E. F., Bigelow, G. E., Johnson, R. E. et al. (2007) QT-interval effects of methadone, levomethadyl, and buprenorphine in a randomized trial. Archives of Internal Medicine 167:2469-2475. doi:10.1001/archinte.167.22.2469
Wolff, K., Rostami‐Hodjegan, A., Hay, A. et al. (2000) Population‐based pharmacokinetic approach for methadone monitoring of opiate addicts: potential clinical utility. Addiction 95(12):1771-1783. doi:10.1046/j.1360-0443.2000.951217717.x
World Health Organization (WHO). (2010). Characterization and application of physiologically based pharmacokinetic models in risk assessment. https://www.inchem.org/documents/harmproj/ harmproj/harmproj9.pdf.
Zhao, S., Kamelia, L., Boonpawa, R. et al. (2019) Physiologically based kinetic modeling-facilitated reverse dosimetry to predict in vivo red blood cell acetylcholinesterase inhibition following exposure to chlorpyrifos in the Caucasian and Chinese population. Toxicological sciences 171(1):69-83. doi:10.1093/toxsci/kfz134
Zwartsen, A., de Korte, T., Nacken, P. et al. (2019) Cardiotoxicity screening of illicit drugs and new psychoactive substances (NPS) in human iPSC-derived cardiomyocytes using microelectrode array (MEA) recordings. Journal of molecular and cellular cardiology 136:102-112. doi:10.1016/j.yjmcc.2019.09.007