U.S. federal agency interests and key considerations for new approach methodologies for nanomaterials
Main Article Content
Abstract
Engineered nanomaterials (ENMs) come in a wide array of shapes, sizes, surface coatings, and compositions, and often possess novel or enhanced properties compared to larger sized particles of the same elemental composition. To ensure the safe commercialization of products containing ENMs, it is important to thoroughly understand their potential risks. Given that ENMs can be created in an almost infinite number of variations, it is not feasible to conduct in vivo testing on each type of ENM. Instead, new approach methodologies (NAMs) such as in vitro or in chemico test methods may be needed, given their capacity for higher throughput testing, lower cost, and ability to provide information on toxicological mechanisms. However, the different behaviors of ENMs compared to dissolved chemicals may challenge safety testing of ENMs using NAMs. In this study, member agencies within the Interagency Coordinating Committee on the Validation of Alternative Methods were queried about what types of ENMs are of agency interest and whether there is agency-specific guidance for ENM toxicity testing. To support the ability of NAMs to provide robust results in ENM testing, two key issues in the usage of NAMs, namely dosimetry and interference/bias controls, are thoroughly discussed.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
U.S.C. § 136 et seq. (1947). Federal Insecticide, Fungicide, and Rodenticide Act. http://www.gpo.gov/fdsys/pkg/USCODE-1996-title7/html/USCODE-1996-title7-chap6-subchapII.htm
U.S.C. § 136 et seq. (1972). Federal Insecticide, Fungicide, and Rodenticide Act (as amended). http://www.gpo.gov/fdsys/pkg/USCODE-1996-title7/html/USCODE-1996-title7-chap6-subchapII.htm
U.S.C. § 136 (1996). Food Quality Protection Act of 1996. govinfo.gov/content/pkg/PLAW-104publ170/pdf/PLAW-104publ170.pdf (accessed 17.10.2019).
U.S.C. § 2601 et seq. (1976). Toxic Substances Control Act. https://www.govinfo.gov/content/pkg/STATUTE-90/pdf/STATUTE-90-Pg2003.pdf#page=1
U.S.C. § 1261 et seq. (2008). Federal Hazardous Substances Act. https://www.govinfo.gov/content/pkg/COMPS-387/pdf/COMPS-387.pdf
U.S.C. § 2601 et seq. (2016). The Frank R. Lautenberg Chemical Safety for the 21st Century Act. https://www.govinfo.gov/content/pkg/PLAW-114publ182/pdf/PLAW-114publ182.pdf
U.S.C. § 301 et seq. (2002). Federal Food, Drug, and Cosmetic Act of 1938 (as amended). https://www.epa.gov/laws-regulations/summary-federal-food-drug-and-cosmetic-act
Adisa, I. O., Reddy Pullagurala, V. L., Rawat, S. et al. (2018). Role of cerium compounds in fusarium wilt suppression and growth enhancement in tomato (Solanum lycopersicum). J Agric Food Chem 66, 5959-5970. doi:10.1021/acs.jafc.8b01345
Amini, R., Brar, S. K., Cledon, M. et al. (2016). Intertechnique comparisons for nanoparticle size measurements and shape distribution. J Hazard, Toxic Radioact Waste 20, B4015004. doi:10.1061/(ASCE)HZ.2153-5515.0000286
Andersen, M. E. and Krewski, D. (2009). Toxicity testing in the 21st century: Bringing the vision to life. J Toxicol Sci 107, 324-330. doi:10.1093/toxsci/kfn255
Armitage, J. M., Wania, F. and Arnot, J. A. (2014). Application of mass balance models and the chemical activity concept to facilitate the use of in vitro toxicity data for risk assessment. Environ Sci Technol 48, 9770-9779. doi:10.1021/es501955g
ASTM E2456-06 (2006). Terminology Relating to Nanotechnology. West Conshohocken, PA: ASTM International. doi:10.1520/E2456-06R12
ASTM E2526-08 (2013). Test Method for Evaluation of Cytotoxicity of Nanoparticulate Materials in Porcine Kidney Cells and Human Hepatocarcinoma Cells. West Conshohocken, PA: ASTM International. doi:10.1520/E2526-08R13
ASTM E2834-12 (2018) (2018). Standard Guide for Measurement of Particle Size Distribution of Nanomaterials in Suspension by Nanoparticle Tracking Analysis (NTA). West Conshohocken, PA: ASTM International. doi:10.1520/E2834-12R18
ASTM E3247-20 (2020). Standard Test Method for Measuring the Size of Nanoparticles in Aqueous Media Using Dynamic Light Scattering. West Conshohocken, PA: ASTM International. doi:10.1520/E3247-20
ASTM E2490-09 (2021). Guide for Measurement of Particle Size Distribution of Nanomaterials in Suspension by Photon Correlation Spectroscopy (PCS). West Conshohocken, PA: ASTM International. doi:10.1520/E2490-09R21
ASTM E3269-21 (2021). Standard Test Method for Determination of the Mass Fraction of Particle-Bound Gold in Colloidal Gold Suspensions. West Conshohocken, PA: ASTM International. doi:10.1520/E3269-21
ASTM WK68060 (2018). New Test Method for Analysis of Liposomal Drug Formulations using Multidetector Asymmetrical-Flow Field-Flow Fractionation (AF4). West Conshohocken, PA: ASTM International. https://www.astm.org/workitem-wk63310 (accessed 21.10.2020).
Bakand, S., Hayes, A. and Dechsakulthorn, F. (2012). Nanoparticles: A review of particle toxicology following inhalation exposure. Inhal Toxicol 24, 125-135. doi:10.3109/08958378.2010.642021
Barosova, H., Maione, A. G., Septiadi, D. et al. (2020). Use of epiAlveolar lung model to predict fibrotic potential of multiwalled carbon nanotubes. ACS Nano 14, 3941-3956. doi:10.1021/acsnano.9b06860
Besinis, A., De Peralta, T., Tredwin, C. J. et al. (2015). Review of nanomaterials in dentistry: Interactions with the oral microenvironment, clinical applications, hazards, and benefits. ACS Nano 9, 2255-2289. doi:10.1021/nn505015e
Bishop, L., Cena, L., Orandle, M. et al. (2017). In vivo toxicity assessment of occupational components of the carbon nanotube life cycle to provide context to potential health effects. ACS Nano 11, 8849-8863. doi:10.1021/acsnano.7b03038
Bohmer, N., Rippl, A., May, S. et al. (2018). Interference of engineered nanomaterials in flow cytometry: A case study. Colloids Surf B Biointerfaces 172, 635-645. doi:10.1016/j.colsurfb.2018.09.021
Borgatta, J., Ma, C., Hudson-Smith, N. et al. (2018). Copper based nanomaterials suppress root fungal disease in watermelon (Citrullus lanatus): Role of particle morphology, composition and dissolution behavior. ACS Sustain Chem Eng 6, 14847-14856. doi:10.1021/acssuschemeng.8b03379
Carvalho, P. M., Felício, M. R., Santos, N. C. et al. (2018). Application of light scattering techniques to nanoparticle characterization and development. Front Chem 6, 237. doi:10.3389/fchem.2018.00237
Casey, W., Chang, X., Allen, D. et al. (2018). Evaluation and optimization of pharmacokinetic models for in vitro to in vivo extrapolation of estrogenic activity for environmental chemicals. Environ Health Perspect 126, 097001. doi:10.1289/EHP1655
CDC/NIOSH (2008). Safe Nanotechnology in the Workplace. An Introduction for Employers, Managers, and Safety and Health Professionals. DHHS (NIOSH) Publication Number 2008-112. https://www.cdc.gov/niosh/docs/2008-112/default.html (accessed 31.12.2020).
CDC/NIOSH (2009a). Approaches to safe Nanotechnology: Managing the Health and Safety Concerns Associated with Engineered Nanomaterials. DHHS (NIOSH) Publication Number 2009-125. https://www.cdc.gov/niosh/docs/2009-125/pdfs/2009-125.pdf
CDC/NIOSH (2009b). Current Intelligence Bulletin 60: Interim Guidance for Medical Screening and Hazard Surveillance for Workers Potentially Exposed to Engineered Nanoparticles. Publication Number 2009-116. https://www.cdc.gov/niosh/docs/2009-116/pdfs/2009-116.pdf
CDC/NIOSH (2011). Current Intelligence Bulletin 63: Occupational Exposure to Titanium Dioxide. Publication Number 2011-160. https://www.cdc.gov/niosh/docs/2011-160/
CDC/NIOSH (2012). General Safe Practices for Working with Engineered Nanomaterials in Research Laboratories. Publication Number 2012-147. https://www.cdc.gov/niosh/docs/2012-147/default.html
CDC/NIOSH (2013). Current Intelligence Bulletin 65: Occupational Exposure to Carbon Nanotubes and Nanofibers. Publication Number 2013-145. https://www.cdc.gov/niosh/docs/2013-145/
CDC/NIOSH (2016). Building a Safety Program to Protect the Nanotechnology Workforce: A Guide for Small to Medium-Sized Enterprises. Publication Number 2016-102. https://www.cdc.gov/niosh/docs/2016-102/default.html (accessed 31.12.2020).
CDC/NIOSH (2018a). Controlling Health Hazards when Working with Nanomaterials: Questions to Ask Before You Start. DHHS (NIOSH) Publication Number 2018-103. https://www.cdc.gov/niosh/docs/2018-103/default.html
CDC/NIOSH (2018b). Workplace Design Solutions: Protecting Workers during Intermediate and Downstream Processing Of Nanomaterials. DHHS (NIOSH) Publication Number 2018-122. https://www.cdc.gov/niosh/docs/2018-122/default.html (accessed 31.12.2020).
CDC/NIOSH (2018c). Workplace Design Solutions: Protecting Workers during Nanomaterial Reactor Operations. DHHS (NIOSH) Publication Number 2018-120. https://www.cdc.gov/niosh/docs/2018-120/default.html (accessed 31.12.2020).
CDC/NIOSH (2018d). Workplace Design Solutions: Protecting Workers during the Handling of Nanomaterials. DHHS (NIOSH) Publication Number 2018-121. https://www.cdc.gov/niosh/docs/2018-121/default.html (accessed 31.12.2020).
Chang, Y., Yang, S.-T., Liu, J.-H. et al. (2011). In vitro toxicity evaluation of graphene oxide on A549 cells. Toxicol Lett 200, 201-210. doi:10.1016/j.toxlet.2010.11.016
Cho, W.-S., Duffin, R., Bradley, M. et al. (2013). Predictive value of in vitro assays depends on the mechanism of toxicity of metal oxide nanoparticles. Part Fibre Toxicol 10, 55. doi:10.1186/1743-8977-10-55
Clippinger, A. J., Raabe, H. A., Allen, D. G. et al. (2021). Human-relevant approaches to assess eye corrosion/irritation potential of agrochemical formulations. Cutan Ocul Toxicol 40, 145-167. doi:10.1080/15569527.2021.1910291
Coyle, J. P., Derk, R. C., Kornberg, T. G. et al. (2020). Carbon nanotube filler enhances incinerated thermoplastics-induced cytotoxicity and metabolic disruption in vitro. Part Fibre Toxicol 17, 40. doi:10.1186/s12989-020-00371-1
CPSC (2019). CPSC Nanomaterial Statement. https://www.cpsc.gov/s3fs-public/CPSCNanostatement.pdf?9SxJOywADgEosXGCgx4N.bgS3X7Skpnu (accessed 08.04.2020).
Davidson, D. C., Derk, R., He, X. et al. (2016). Direct stimulation of human fibroblasts by nCeO2 in vitro is attenuated with an amorphous silica coating. Part Fibre Toxicol 13, 23. doi:10.1186/s12989-016-0134-8
DeLoid, G., Cohen, J. M., Darrah, T. et al. (2014). Estimating the effective density of engineered nanomaterials for in vitro dosimetry. Nat Commun 5, 3514. doi:10.1038/ncomms4514
DeLoid, G. M., Cohen, J. M., Pyrgiotakis, G. et al. (2017). Preparation, characterization, and in vitro dosimetry of dispersed, engineered nanomaterials. Nat Protoc 12, 355-371. doi:10.1038/nprot.2016.172
Drasler, B., Sayre, P., Steinhäuser, K. G. et al. (2017). In vitro approaches to assess the hazard of nanomaterials. NanoImpact 8, 99-116. doi:10.1016/j.impact.2017.08.002
Edgington, A., Petersen, E. J., Herzing, A. A. et al. (2014). Microscopic investigation of single-wall carbon nanotube uptake by Daphnia magna. Nanotoxicology 8, 2-10. doi:10.3109/17435390.2013.847504
Elliott, J. T., Rösslein, M., Song, N. W. et al. (2017). Toward achieving harmonization in a nano-cytotoxicity assay measurement through an interlaboratory comparison study. ALTEX 34, 201-218. doi:10.14573/altex.1605021
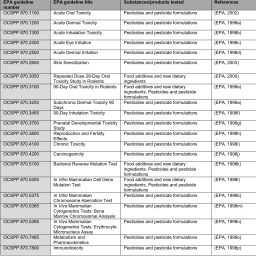
EPA (1996a). Health Effects Test Guidelines: OCSPP 870.3250 – Subchronic Dermal Toxicity 90 days. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100IRLD.PDF?Dockey=P100IRLD.PDF
EPA (1996b). Health Effects Test Guidelines: OCSPP 870.5375 – In Vitro Mammalian Chromosome Aberration Test. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100ID5Q.PDF?Dockey=P100ID5Q.PDF
EPA (1998a). Health Effects Test Guidelines: OCSPP 870.1200 – Acute Dermal Toxicity. Washington, DC: U.S. Environmental Protection Agency. http://www.epa.gov.opptsfrs/publications/OPPTS_Harmonized/870_Health_Effects_Test_Guidelines/Series/870-1200.pdf
EPA (1998b). Health Effects Test Guidelines: OCSPP 870.1300 – Acute Inhalation Toxicity. Washington, DC: U.S. Environmental Protection Agency. https://ntp.niehs.nih.gov/iccvam/suppdocs/feddocs/epa/epa-870-1300.pdf
EPA (1998c). Health Effects Test Guidelines: OCSPP 870.2400 – Acute Eye Irritation. Washington, DC: U.S. Environmental Protection Agency. https://www.regulations.gov/document?D=EPA-HQ-OPPT-2009-0156-0006
EPA (1998d). Health Effects Test Guidelines: OCSPP 870.2500 – Acute Dermal Irritation. Washington, DC: U.S. Environmental Protection Agency. https://ntp.niehs.nih.gov/iccvam/suppdocs/feddocs/epa/epa_870_2500.pdf
EPA (1998e). Health Effects Test Guidelines: OCSPP 870.3100 – 90-Day Oral Toxicity in Rodents. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100ID3R.PDF?Dockey=P100ID3R.PDF (accessed 26.03.2020).
EPA (1998f). Health Effects Test Guidelines: OCSPP 870.3465 – 90-Day Inhalation Toxicity. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100IRTA.PDF?Dockey=P100IRTA.PDF
EPA (1998g). Health Effects Test Guidelines: OCSPP 870.3700 – Prenatal Developmental Toxicity Study. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100ID44.PDF?Dockey=P100ID44.PDF
EPA (1998h). Health Effects Test Guidelines: OCSPP 870.3800 – Reproduction and Fertility Effects. Washington, DC: U.S. Environmental Protection Agency. https://www.epa.gov/test-guidelines-pesticides-and-toxic-substances/series-870-health-effects-test-guidelines
EPA (1998i). Health Effects Test Guidelines: OCSPP 870.4100 – Chronic Toxicity. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100IRTR.PDF?Dockey=P100IRTR.PDF
EPA (1998j). Health Effects Test Guidelines: OCSPP 870.4200 – Carcinogenicity. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100J73B.PDF?Dockey=P100J73B.PDF
EPA (1998k). Health Effects Test Guidelines: OCSPP 870.5100 – Bacterial Reverse Mutation Test. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100ID6E.PDF?Dockey=P100ID6E.PDF
EPA (1998l). Health Effects Test Guidelines: OCSPP 870.5300 – In Vitro Mammalian Cell Gene Mutation Test. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100ID5C.PDF?Dockey=P100ID5C.PDF
EPA (1998m). Health Effects Test Guidelines: OCSPP 870.5385 – In Vivo Mammalian Cytogenetics Tests: Bone Marrow Chromosomal Analysis. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100A209.PDF?Dockey=P100A209.PDF
EPA (1998n). Health Effects Test Guidelines: OCSPP 870.5395 – In Vivo Mammalian Cytogenetics Tests: Erythrocyte Micronucleus Assay. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100A20G.PDF?Dockey=P100A20G.PDF
EPA (1998o). Health Effects Test Guidelines: OCSPP 870.7485 – Metabolism and Pharmacokinetics. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100IRJI.PDF?Dockey=P100IRJI.PDF
EPA (1998p). Health Effects Test Guidelines: OCSPP 870.7800 – Immunotoxicity. Washington, DC: U.S. Environmental Protection Agency. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100IRS7.PDF?Dockey=P100IRS7.PDF
EPA (2000). Health Effects Test Guidelines: OCSPP 870.3050 – Repeated Dose 28-Day Oral Toxicity Study in Rodents. Washington, DC: U.S. Environmental Protection Agency. https://ntp.niehs.nih.gov/iccvam/suppdocs/feddocs/epa/epa_870_3050.pdf
EPA (2002). Health Effects Test Guidelines: OCSPP 870.1100 – Acute Oral Toxicity. Washington, DC: U.S. Environmental Protection Agency. https://www.epa.gov/test-guidelines-pesticides-and-toxic-substances/series-870-health-effects-test-guidelines (accessed 19.03.2019).
EPA (2003). Health Effects Test Guidelines: OCSPP 870.2600 – Skin Sensitization. Washington, DC: U.S. Environmental Protection Agency. http://www.regulations.gov/#!documentDetail;D=EPA-HQ-OPPT-2009-0156-0008 (accessed 17.09.2009).
EPA (2017). Working Guidance on EPA’s Section 8(a) Information Gathering Rule on Nanomaterials in Commerce. https://www.epa.gov/sites/production/files/2017-08/documents/august_2017guidance.8-7-2017_002.pdf (accessed 08.04.2020).
EPA (2018). Strategic Plan to Promote the Development and Implementation of Alternative Test Methods Within the TSCA Program. EPA document# EPA-740-R1-8004. https://www.epa.gov/sites/production/files/2018-06/documents/epa_alt_strat_plan_6-20-18_clean_final.pdf (accessed 23.07.2020).
EPA (2021). List of Alternative Test Methods and Strategies (or New Approach Methodologies [NAMs]). https://www.epa.gov/sites/production/files/2021-02/documents/nams_list_second_update_2-4-21_final.pdf (accessed 30.04.2021).
Fadeel, B., Bussy, C., Merino, S. et al. (2018). Safety assessment of graphene-based materials: Focus on human health and the environment. ACS Nano 12, 10582-10620. doi:10.1021/acsnano.8b04758.
Farjadian, F., Ghasemi, A., Gohari, O. et al. (2019). Nanopharmaceuticals and nanomedicines currently on the market: challenges and opportunities. Nanomedicine (Lond) 14, 93-126. doi:10.2217/nnm-2018-0120
FDA (2007). Nanotechnology: A Report of the U.S. Food and Drug Administration Nanotechnology Task Force. https://www.fda.gov/media/74257/download (accessed 05.05.2020).
FDA (2014a). Guidance for Industry: Assessing the Effects of Significant Manufacturing Process Changes, Including Emerging Technologies, on the Safety and Regulatory Status of Food Ingredients and Food Contact Substances, Including Food Ingredients that are Color Additives. https://www.fda.gov/media/115075/download
FDA (2014b). Guidance for Industry: Considering Whether an FDA-Regulated Product Involves the Application of Nanotechnology. https://www.fda.gov/media/88423/download (accessed 08.04.2020).
FDA (2014c). Guidance for Industry: Safety of Nanomaterials in Cosmetic Products. https://www.fda.gov/media/83957/download
FDA (2015). Guidance for Industry: Use of Nanomaterials in Food for Animals. https://www.fda.gov/media/88828/download
FDA (2017). Draft Guidance for Industry: Drug Products, Including Biological Products, that Contain Nanomaterials. Please note, this document is draft and not for implementation. Once finalized, the document will represent the FDA’s position. https://www.fda.gov/media/109910/download (accessed 19.03.2021).
FDA (2020). Nanotechnology – Over a Decade of Progress and Innovation. A report by the U.S. food and drug administration. https://www.fda.gov/media/140395/download
Fenech, M. (1997). The advantages and disadvantages of the cytokinesis-block micronucleus method. Mutat Res 392, 11-18. doi:10.1016/S0165-1218(97)00041-4
Geitner, N. K., Hendren, C. O., Cornelis, G. et al. (2020). Harmonizing across environmental nanomaterial testing media for increased comparability of nanomaterial datasets. Environ Sci Nano 7, 13-36. doi:10.1039/C9EN00448C
Gerloff, K., Landesmann, B., Worth, A. et al. (2017). The adverse outcome pathway approach in nanotoxicology. Comput Toxicology 1, 3-11. doi:10.1016/j.comtox.2016.07.001
Gonzalez, L., Sanderson, B. J. S. and Kirsch-Volders, M. (2011). Adaptations of the in vitro MN assay for the genotoxicity assessment of nanomaterials. Mutagenesis 26, 185-191. doi:10.1093/mutage/geq088
Goodwin, D. G., Adeleye, A. S., Sung, L. et al. (2018). Detection and quantification of graphene-family nanomaterials in the environment. Environ Sci Technol 52, 4491-4513. doi:10.1021/acs.est.7b04938
Grafmueller, S., Manser, P., Diener, L. et al. (2015). Bidirectional transfer study of polystyrene nanoparticles across the placental barrier in an ex vivo human placental perfusion model. Environ Health Perspect 123, 1280-1286. doi:10.1289/ehp.1409271
Grieger, K. D., Hansen, S. F. and Baun, A. (2009). The known unknowns of nanomaterials: Describing and characterizing uncertainty within environmental, health and safety risks. Nanotoxicology 3, 222-233. doi:10.1080/17435390902944069
Griffitt, R. J., Luo, J., Gao, J. et al. (2008). Effects of particle composition and species on toxicity of metallic nanomaterials in aquatic organisms. Environ Toxicol Chem 27, 1972. doi:10.1897/08-002.1
Guadagnini, R., Halamoda Kenzaoui, B., Walker, L. et al. (2015). Toxicity screenings of nanomaterials: Challenges due to interference with assay processes and components of classic in vitro tests. Nanotoxicology 9, 13-24. doi:10.3109/17435390.2013.829590
Ha, M. K., Shim, Y. J. and Yoon, T. H. (2018). Effects of agglomeration on in vitro dosimetry and cellular association of silver nanoparticles. Environ Sci Nano 5, 446-455. doi:10.1039/C7EN00965H
Hadrup, N., Zhernovkov, V., Jacobsen, N. R. et al. (2020). Acute phase response as a biological mechanism-of-action of (nano)particle-induced cardiovascular disease. Small 16, 1907476. doi:10.1002/smll.201907476
Halappanavar, S., Ede, J. D., Shatkin, J. A. et al. (2019). A systematic process for identifying key events for advancing the development of nanomaterial relevant adverse outcome pathways. NanoImpact 15, 100178. doi:10.1016/j.impact.2019.100178
Halappanavar, S., Ede, J. D., Mahapatra, I. et al. (2020). A methodology for developing key events to advance nanomaterial-relevant adverse outcome pathways to inform risk assessment. Nanotoxicology 15, 289-310. doi:10.1080/17435390.2020.1851419
Han, X., Gelein, R., Corson, N. et al. (2011). Validation of an LDH assay for assessing nanoparticle toxicity. Toxicology 287, 99-104. doi:10.1016/j.tox.2011.06.011
Hanna, S. K., Cooksey, G. A., Dong, S. et al. (2016). Feasibility of using a standardized Caenorhabditis elegans toxicity test to assess nanomaterial toxicity. Environ Sci Nano 3, 1080-1089. doi:10.1039/C6EN00105J
Hanna, S. K., Bustos, A. R. M., Peterson, A. W. et al. (2018). Agglomeration of Escherichia coli with positively charged nanoparticles can lead to artifacts in a standard Caenorhabditis elegans toxicity assay. Environ Sci Technol 52, 5968-5978. doi:10.1021/acs.est.7b06099
Haque, E. and Ward, A. C. (2018). Zebrafish as a model to evaluate nanoparticle toxicity. Nanomaterials (Basel) 8, 561. doi:10.3390/nano8070561
Harper, S., Usenko, C., Hutchison, J. E. et al. (2008). In vivo biodistribution and toxicity depends on nanomaterial composition, size, surface functionalisation and route of exposure. J Exp Nanosci 3, 195-206. doi:10.1080/17458080802378953
Hartmann, N. B., Engelbrekt, C., Zhang, J. et al. (2012). The challenges of testing metal and metal oxide nanoparticles in algal bioassays: Titanium dioxide and gold nanoparticles as case studies. Nanotoxicology 7, 1082-1094. doi:10.3109/17435390.2012.710657
Hinderliter, P. M., Minard, K. R., Orr, G. et al. (2010). ISDD: A computational model of particle sedimentation, diffusion and target cell dosimetry for in vitro toxicity studies. Part Fibre Toxicol 7, 36. doi:10.1186/1743-8977-7-36
Holder, A. L., Goth-Goldstein, R., Lucas, D. et al. (2012). Particle-induced artifacts in the MTT and LDH viability assays. Chem Res Toxicol 25, 1885-1892. doi:10.1021/tx3001708
Horst, A. M., Vukanti, R., Priester, J. H. et al. (2013). An assessment of fluorescence- and absorbance-based assays to study metal-oxide nanoparticle ROS production and effects on bacterial membranes. Small 9, 1753-1764. doi:10.1002/smll.201201455
ICCVAM (2018). A Strategic Roadmap for Establishing New Approaches to Evaluate the Safety of Chemicals and Medical Products in the United States. Research Triangle Park, NC: National Institute of Environmental Health Sciences. doi:10.22427/NTP-ICCVAM-ROADMAP2018
ISO (2016). ISO 19430:2016 Particle Size Analysis – Particle Tracking Analysis (PTA) Method. https://www.iso.org/cms/render/live/en/sites/isoorg/contents/data/standard/06/48/64890.html (accessed 13.08.2021).
ISO (2019). ISO 20814:2019 Nanotechnologies – Testing the Photocatalytic Activity of Nanoparticles for NADH Oxidation. https://www.iso.org/standard/69298.html
ISO (2020a). ISO 17200:2020 Nanotechnology – Nanoparticles in Powder Form – Characteristics and Measurements. https://www.iso.org/cms/render/live/en/sites/isoorg/contents/data/standard/06/36/63698.html (accessed 21.10.2020).
ISO (2020b). ISO 17867:2020 Particle Size Analysis – Small Angle X-ray Scattering (SAXS). https://www.iso.org/cms/render/live/en/sites/isoorg/contents/data/standard/06/92/69213.html (accessed 13.08.2021).
ISO (2020c). ISO 21363:2020 Nanotechnologies – Measurements of Particle Size and Shape Distributions by Transmission Electron Microscopy. https://www.iso.org/cms/render/live/en/sites/isoorg/contents/data/standard/07/07/70762.html (accessed 13.08.2021).
ISO (2020d). ISO/TR 21624:2020 Nanotechnologies – Considerations for In vitro Studies of Airborne Nano‐Objects and their Aggregates and Agglomerates (NOAA). https://www.iso.org/cms/render/live/en/sites/isoorg/contents/data/standard/07/12/71273.html (accessed 04.06.2020).
ISO (2021). ISO 19749:2021 Nanotechnologies – Measurements of Particle Size and Shape Distributions by Scanning Electron Microscopy (SEM). https://www.iso.org/cms/render/live/en/sites/isoorg/contents/data/standard/06/48/64890.html (accessed 13.08.2021).
Jeevanandam, J., Barhoum, A., Chan, Y. S. et al. (2018). Review on nanoparticles and nanostructured materials: History, sources, toxicity and regulations. Beilstein J Nanotechnol 9, 1050-1074. doi:10.3762/bjnano.9.98
Johnston, L. J., Gonzalez-Rojano, N., Wilkinson, K. J. et al. (2020). Key challenges for evaluation of the safety of engineered nanomaterials. NanoImpact 18, 100219. doi:10.1016/j.impact.2020.100219
Kah, M., Tufenkji, N. and White, J. C. (2019). Nano-enabled strategies to enhance crop nutrition and protection. Nat Nanotechnol 14, 532-540. doi:10.1038/s41565-019-0439-5
Kaiser, J.-P., Roesslein, M., Diener, L. et al. (2017). Cytotoxic effects of nanosilver are highly dependent on the chloride concentration and the presence of organic compounds in the cell culture media. J Nanobiotechnology 15, 5. doi:10.1186/s12951-016-0244-3
Keene, A. M., Bancos, S. and Tyner, K. M. (2014). Considerations for in vitro nanotoxicity testing. In Handbook of Nanotoxicology, Nanomedicine and Stem Cell Use in Toxicology (35-64). John Wiley & Sons, Ltd. doi:10.1002/9781118856017.ch2
Kettiger, H., Schipanski, A., Wick, P. et al. (2013). Engineered nanomaterial uptake and tissue distribution: From cell to organism. Int J Nanomedicine 8, 3255-3269. doi:10.2147/IJN.S49770
Kim, Y., Yoon, C., Ham, S. et al. (2015). Emissions of nanoparticles and gaseous material from 3D printer operation. Environ Sci Technol 49, 12044-12053. doi:10.1021/acs.est.5b02805
Kobets, T., Iatropoulos, M. J. and Williams, G. M. (2018). Mechanisms of DNA-reactive and epigenetic chemical carcinogens: Applications to carcinogenicity testing and risk assessment. Toxicol Res (Camb) 8, 123-145. doi:10.1039/c8tx00250a
Kodali, V. K., Roberts, J. R., Shoeb, M. et al. (2017). Acute in vitro and in vivo toxicity of a commercial grade boron nitride nanotube mixture. Nanotoxicology 11, 1040-1058. doi:10.1080/17435390.2017.1390177
Krewski, D., Westphal, M., Andersen, M. E. et al. (2014). A framework for the next generation of risk science. Environ Health Perspect 122, 796-805. doi:10.1289/ehp.1307260
Kroll, A., Pillukat, M. H., Hahn, D. et al. (2012). Interference of engineered nanoparticles with in vitro toxicity assays. Arch Toxicol 86, 1123-1136. doi:10.1007/s00204-012-0837-z
Kühnel, D. and Nickel, C. (2014). The OECD expert meeting on ecotoxicology and environmental fate – Towards the development of improved OECD guidelines for the testing of nanomaterials. Sci Total Environ 472, 347-353. doi:10.1016/j.scitotenv.2013.11.055
Laborda, F., Bolea, E., Cepriá, G. et al. (2016). Detection, characterization and quantification of inorganic engineered nanomaterials: A review of techniques and methodological approaches for the analysis of complex samples. Anal Chim Acta 904, 10-32. doi:10.1016/j.aca.2015.11.008
Lacroix, G., Koch, W., Ritter, D. et al. (2018). Air-liquid interface in vitro models for respiratory toxicology research: Consensus workshop and recommendations. Appl In Vitro Toxicol 4, 91-106. doi:10.1089/aivt.2017.0034
Lead, J. R., Batley, G. E., Alvarez, P. J. J. et al. (2018). Nanomaterials in the environment: Behavior, fate, bioavailability, and effects – An updated review. Environ Toxicol Chem 37, 2029-2063. doi:10.1002/etc.4147
Leibrock, L. B., Jungnickel, H., Tentschert, J. et al. (2020). Parametric optimization of an air-liquid interface system for flow-through inhalation exposure to nanoparticles: Assessing dosimetry and intracellular uptake of CeO2 nanoparticles. Nanomaterials 10, 2369. doi:10.3390/nano10122369
Li, X., Lenhart, J. J. and Walker, H. W. (2010). Dissolution-accompanied aggregation kinetics of silver nanoparticles. Langmuir 26, 16690-16698. doi:10.1021/la101768n
Lin, M.-H., Hsu, T.-S., Yang, P.-M. et al. (2009). Comparison of organic and inorganic germanium compounds in cellular radiosensitivity and preparation of germanium nanoparticles as a radiosensitizer. Int J Radiat Biol 85, 214-226. doi:10.1080/09553000902748583
Liu, J., Sonshine, D. A., Shervani, S. et al. (2010). Controlled release of biologically active silver from nanosilver surfaces. ACS Nano 4, 6903-6913. doi:10.1021/nn102272n
Lowry, G. V., Avellan, A. and Gilbertson, L. M. (2019). Opportunities and challenges for nanotechnology in the agri-tech revolution. Nat Nanotechnol 14, 517-522. doi:10.1038/s41565-019-0461-7
Luechtefeld, T., Maertens, A., Russo, D. P. et al. (2016). Global analysis of publicly available safety data for 9,801 substances registered under REACH from 2008-2014. ALTEX 33, 95-109. doi:10.14573/altex.1510052
Ma, J., Mercer, R. R., Barger, M. et al. (2015). Effects of amorphous silica coating on cerium oxide nanoparticles induced pulmonary responses. Toxicol Appl Pharmacol 288, 63-73. doi:10.1016/j.taap.2015.07.012
MacLean-Fletcher, S. and Pollard, T. D. (1980). Mechanism of action of cytochalasin B on actin. Cell 20, 329-341. doi:10.1016/0092-8674(80)90619-4
Manke, A., Luanpitpong, S., Dong, C. et al. (2014). Effect of fiber length on carbon nanotube-induced fibrogenesis. Int J Mol Sci 15, 7444-7461. doi:10.3390/ijms15057444
Mercer, R. R., Hubbs, A. F., Scabilloni, J. F. et al. (2011). Pulmonary fibrotic response to aspiration of multi-walled carbon nanotubes. Part Fibre Toxicol 8, 21. doi:10.1186/1743-8977-8-21
Minelli, C., Bartczak, D., Peters, R. et al. (2019). Sticky measurement problem: Number concentration of agglomerated nanoparticles. Langmuir 35, 4927-4935. doi:10.1021/acs.langmuir.8b04209
Mishra, A., Rojanasakul, Y., Chen, B. T. et al. (2012). Assessment of pulmonary fibrogenic potential of multiwalled carbon nanotubes in human lung cells. J Nanomater 2012, e930931. doi:10.1155/2012/930931
Mo, J., Xie, Q., Wei, W. et al. (2018). Revealing the immune perturbation of black phosphorus nanomaterials to macrophages by understanding the protein corona. Nat Commun 9, 2480. doi:10.1038/s41467-018-04873-7
Monteiro-Riviere, N. A., Inman, A. O. and Zhang, L. W. (2009). Limitations and relative utility of screening assays to assess engineered nanoparticle toxicity in a human cell line. Toxicol Appl Pharmacol 234, 222-235. doi:10.1016/j.taap.2008.09.030
Mortimer, M., Petersen, E. J., Buchholz, B. A. et al. (2016). Bioaccumulation of multiwall carbon nanotubes in tetrahymena thermophila by direct feeding or trophic transfer. Environ Sci Technol 50, 8876-8885. doi:10.1021/acs.est.6b01916
Mourdikoudis, S., Pallares, R. M. and Thanh, N. T. K. (2018). Characterization techniques for nanoparticles: Comparison and complementarity upon studying nanoparticle properties. Nanoscale 10, 12871-12934. doi:10.1039/C8NR02278J
Natsch, A., Laue, H., Haupt, T. et al. (2018). Accurate prediction of acute fish toxicity of fragrance chemicals with the RTgill-W1 cell assay. Environ Toxicol Chem 37, 931-941. doi:10.1002/etc.4027
Nel, A., Nasser, E., Godwin, H. et al. (2013a). A multi-stakeholder perspective on the use of alternative test strategies for nanomaterial safety assessment. ACS Nano 7, 6422-6433. doi:10.1021/nn4037927
Nel, A., Xia, T., Meng, H. et al. (2013b). Nanomaterial toxicity testing in the 21st century: Use of a predictive toxicological approach and high-throughput screening. Acc Chem Res 46, 607-621. doi:10.1021/ar300022h
Nelson, B. C., Petersen, E. J., Marquis, B. J. et al. (2013). NIST gold nanoparticle reference materials do not induce oxidative DNA damage. Nanotoxicology 7, 21-29. doi:10.3109/17435390.2011.626537
NRC (2007). Toxicity Testing in the 21st Century: A Vision and a Strategy. Washington, DC, USA: National Academies Press. http://books.nap.edu/openbook.php?record_id=11970
Oberdörster, G., Oberdörster, E. and Oberdörster, J. (2005). Nanotoxicology: An emerging discipline evolving from studies of ultrafine particles. Environ Health Perspect 113, 823-839. doi:10.1289/ehp.7339
OECD (1998a). Test No. 212: Fish, Short-term Toxicity Test on Embryo and Sac-fry Stages. OECD Guidelines for the Testing of Chemicals, Section 2. OECD Publishing, Paris. https://www.oecd-ilibrary.org/content/publication/9789264070141-en
OECD (1998b). Test No. 408: Repeated Dose 90-day Oral Toxicity Study in Rodents. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. doi:10.1787/9789264070707-en
OECD (2008). Test No. 407: Repeated Dose 28-day Oral Toxicity Study in Rodents. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. doi:10.1787/9789264070684-en
OECD (2009a). Preliminary Review of OECD Test Guidelines for their Applicability to Manufactured Nanomaterials. Series on the Safety of Manufactured Nanomaterials No. 15. http://www.oecd.org/officialdocuments/publicdisplaydocumentpdf/?doclanguage=en&cote=env/jm/mono(2009)21 (accessed 27.08.2020).
OECD (2009b). Test No. 412: Subacute Inhalation Toxicity: 28-Day Study. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. doi:10.1787/9789264070783-en
OECD (2012). Inhalation Toxicity Testing: Expert Meeting on Potential Revisions to OECD Test Guidelines and Guidance Document. Series on the Safety of Manufactured Nanomaterials No. 35. http://www.oecd.org/officialdocuments/publicdisplaydocumentpdf/?cote=env/jm/mono(2012)14&doclanguage=en (accessed 27.08.2020).
OECD (2016). Test No. 487: In Vitro Mammalian Cell Micronucleus Test. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. doi:10.1787/9789264264861-en
OECD (2018a). Evaluation of In Vitro Methods For Human Hazard Assessment Applied in the OECD Testing Programme for the Safety of Manufactured Nanomaterials. Series on the Safety of Manufactured Nanomaterials No. 85. http://www.oecd.org/officialdocuments/publicdisplaydocumentpdf/?cote=ENV/JM/MONO%282018%294&doclanguage=en
OECD (2018b). Test No. 413: Subchronic Inhalation Toxicity: 90-day Study. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. https://doi.org/10.1787/9789264070806-en.
OECD (2018c). Test No. 442D: In Vitro Skin Sensitisation: ARE-Nrf2 Luciferase Test Method. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. doi:10.1787/9789264229822-en
OECD (2019). Test No. 442C: In Chemico Skin Sensitisation: Assays Addressing the Adverse Outcome Pathway Key Event on Covalent Binding to Proteins. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. doi:10.1787/9789264229709-en
OECD (2020). OECD Series on Testing and Assessment No. 317: Guidance Document on Aquatic and Sediment Toxicity Testing of Nanomaterials. Series on Testing and Assessment No. 317. OECD Publishing, Paris. http://www.oecd.org/officialdocuments/publicdisplaydocumentpdf/?cote=env/jm/mono(2020)8&doclanguage=en (accessed 23.10.2020).
OECD (2021a). Test No. 439: In Vitro Skin Irritation: Reconstructed Human Epidermis Test Method. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. doi:10.1787/9789264242845-en
OECD (2021b). Guideline No. 497: Defined Approaches on Skin Sensitisation. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. https://doi.org/10.1787/b92879a4-en
Oischinger, J., Meiller, M., Daschner, R. et al. (2019). Fate of nano titanium dioxide during combustion of engineered nanomaterial-containing waste in a municipal solid waste incineration plant. Waste Manag Res 37, 1033-1042. doi:10.1177/0734242X19862603
Ong, K. J., MacCormack, T. J., Clark, R. J. et al. (2014). Widespread nanoparticle-assay interference: Implications for nanotoxicity testing. PLoS One 9, e90650. doi:10.1371/journal.pone.0090650
Pal, A. K., Bello, D., Cohen, J. et al. (2015). Implications of in-vitro dosimetry on toxicological ranking of low aspect ratio engineered nanomaterials. Nanotoxicology 9, 871-885. doi:10.3109/17435390.2014.986670
Petersen, E. J., Pinto, R. A., Mai, D. J. et al. (2011). Influence of polyethyleneimine graftings of multi-walled carbon nanotubes on their accumulation and elimination by and toxicity to Daphnia magna. Environ Sci Technol 45, 1133-1138. doi:10.1021/es1030239
Petersen, E. J. and Henry, T. B. (2012). Methodological considerations for testing the ecotoxicity of carbon nanotubes and fullerenes: Review. Environ Toxicol Chem 31, 60-72. doi:10.1002/etc.710
Petersen, E. J., Pinto, R. A., Shi, X. et al. (2012). Impact of size and sorption on degradation of trichloroethylene and polychlorinated biphenyls by nano-scale zerovalent iron. J Hazard Mater 243, 73-79. doi:10.1016/j.jhazmat.2012.09.070
Petersen, E. J., Henry, T. B., Zhao, J. et al. (2014). Identification and avoidance of potential artifacts and misinterpretations in nanomaterial ecotoxicity measurements. Environ Sci Technol 48, 4226-4246. doi:10.1021/es4052999
Petersen, E. J., Flores-Cervantes, D. X., Bucheli, T. D. et al. (2016). Quantification of carbon nanotubes in environmental matrices: Current capabilities, case studies, and future prospects. Environ Sci Technol 50, 4587-4605. doi:10.1021/acs.est.5b05647
Petersen, E. J., Bustos, A. R. M., Toman, B. et al. (2019a). Determining what really counts: Modeling and measuring nanoparticle number concentrations. Environ Sci Nano 6, 2876-2896. doi:10.1039/C9EN00462A
Petersen, E. J., Mortimer, M., Burgess, R. M. et al. (2019b). Strategies for robust and accurate experimental approaches to quantify nanomaterial bioaccumulation across a broad range of organisms. Environ Sci Nano 6, 1619-1656. doi:10.1039/C8EN01378K
Petersen, E. J., Hirsch, C., Elliott, J. T. et al. (2020). Cause-and-effect analysis as a tool to improve the reproducibility of nanobioassays: Four case studies. Chem Res Toxicol 33, 1039-1054. doi:10.1021/acs.chemrestox.9b00165
Pham, L., Watford, S., Pradeep, P. et al. (2020). Variability in in vivo studies: Defining the upper limit of performance for predictions of systemic effect levels. Comput Toxicol 15, 1-100126. doi:10.1016/j.comtox.2020.100126
Pirela, S. V., Bhattacharya, K., Wang, Y. et al. (2019). A 21-day sub-acute, whole-body inhalation exposure to printer-emitted engineered nanoparticles in rats: Exploring pulmonary and systemic effects. NanoImpact 15, 100176. doi:10.1016/j.impact.2019.100176
Poli, D., Mattei, G., Ucciferri, N. et al. (2020). An integrated in vitro-in silico approach for silver nanoparticle dosimetry in cell cultures. Ann Biomed Eng 48, 1271-1280. doi:10.1007/s10439-020-02449-5
Polk, W. W., Sharma, M., Sayes, C. M. et al. (2016). Aerosol generation and characterization of multi-walled carbon nanotubes exposed to cells cultured at the air-liquid interface. Part Fibre Toxicol 13, 20. doi:10.1186/s12989-016-0131-y
Price, S. R., Kinnear, C. and Balog, S. (2019). Particokinetics and in vitro dose of high aspect ratio nanoparticles. Nanoscale 11, 5209-5214. doi:10.1039/C9NR00976K
Rampado, R., Crotti, S., Caliceti, P. et al. (2020). Recent advances in understanding the protein corona of nanoparticles and in the formulation of “stealthy” nanomaterials. Front Bioeng Biotechnol 8, 166. doi:10.3389/fbioe.2020.00166
Renero-Lecuna, C., Iturrioz-Rodríguez, N., González-Lavado, E. et al. (2019). Effect of size, shape, and composition on the interaction of different nanomaterials with HeLa cells. J Nanomater 2019, e7518482. doi:10.1155/2019/7518482
Rice, P. A., Cassidy, K. S., Mihalov, J. et al. (2009). Safety assessment of engineered nanomaterials in direct food additives and food contact materials. In S. C. Sahu and D. A. Casciano (eds.), Nanotoxicity (581-595). John Wiley & Sons, Ltd. doi:10.1002/9780470747803.ch26
Ridge, S. J. (2018). A Regulatory Framework for Nanotechnology. Monterery, CA, USA: Naval Postgraduate School. https://www.hsaj.org/articles/14554 (accessed 09.08.2021).
Roberts, J. R., Antonini, J. M., Porter, D. W. et al. (2013). Lung toxicity and biodistribution of Cd/Se-ZnS quantum dots with different surface functional groups after pulmonary exposure in rats. Part Fibre Toxicol 10, 5. doi:10.1186/1743-8977-10-5
Rooney, J. P., Choksi, N. Y., Ceger, P. et al. (2021). Analysis of variability in the rabbit skin irritation assay. Regul Toxicol Pharmacol 122, 104920. doi:10.1016/j.yrtph.2021.104920
Rösslein, M., Hirsch, C., Kaiser, J.-P. et al. (2013). Comparability of in vitro tests for bioactive nanoparticles: A common assay to detect reactive oxygen species as an example. Int J Mol Sci 14, 24320-24337. doi:10.3390/ijms141224320
Rösslein, M., Elliott, J. T., Salit, M. et al. (2015). Use of cause-and-effect analysis to design a high-quality nanocytotoxicology assay. Chem Res Toxicol 28, 21-30. doi:10.1021/tx500327y
Rösslein, M., Liptrott, N. J., Owen, A. et al. (2017). Sound understanding of environmental, health and safety, clinical, and market aspects is imperative to clinical translation of nanomedicines. Nanotoxicology 11, 147-149. doi:10.1080/17435390.2017.1279361
Rothen-Rutishauser, B., Mühlfeld, C., Blank, F. et al. (2007). Translocation of particles and inflammatory responses after exposure to fine particles and nanoparticles in an epithelial airway model. Part Fibre Toxicol 4, 9. doi:10.1186/1743-8977-4-9
Saber, A. T., Jacobsen, N. R., Jackson, P. et al. (2014). Particle-induced pulmonary acute phase response may be the causal link between particle inhalation and cardiovascular disease. Wiley Interdiscip Rev Nanomed Nanobiotechnol 6, 517-531. doi:10.1002/wnan.1279
Sager, T. M. and Castranova, V. (2009). Surface area of particle administered versus mass in determining the pulmonary toxicity of ultrafine and fine carbon black: Comparison to ultrafine titanium dioxide. Part Fibre Toxicol 6, 15. doi:10.1186/1743-8977-6-15
Salieri, B., Kaiser, J.-P., Rösslein, M. et al. (2020). Relative potency factor approach enables the use of in vitro information for estimation of human effect factors for nanoparticle toxicity in life-cycle impact assessment. Nanotoxicology 14, 275-286. doi:10.1080/17435390.2019.1710872
Sargent, L. M., Porter, D. W., Staska, L. M. et al. (2014). Promotion of lung adenocarcinoma following inhalation exposure to multi-walled carbon nanotubes. Part Fibre Toxicol 11, 3. doi:10.1186/1743-8977-11-3
Scanlan, L. D., Reed, R. B., Loguinov, A. V. et al. (2013). Silver nanowire exposure results in internalization and toxicity to Daphnia magna. ACS Nano 7, 10681-10694. doi:10.1021/nn4034103
Shaffer, R. M. (2021). Environmental health risk assessment in the federal government: A visual overview and a renewed call for coordination. Environ Sci Technol 55, 10923-10927. doi:10.1021/acs.est.1c01955
Shatkin, J. A., Ong, K. J., Beaudrie, C. et al. (2016). Advancing risk analysis for nanoscale materials: Report from an international workshop on the role of alternative testing strategies for advancement. Risk Anal 36, 1520-1537. doi:10.1111/risa.12683
Siegrist, K. J., Reynolds, S. H., Kashon, M. L. et al. (2014). Genotoxicity of multi-walled carbon nanotubes at occupationally relevant doses. Part Fibre Toxicol 11, 6. doi:10.1186/1743-8977-11-6
Snyder-Talkington, B. N., Dong, C., Zhao, X. et al. (2015). Multi-walled carbon nanotube-induced gene expression in vitro: Concordance with in vivo studies. Toxicology 328, 66-74. doi:10.1016/j.tox.2014.12.012
Snyder-Talkington, B. N., Dong, C., Castranova, V. et al. (2019). Differential gene regulation in human small airway epithelial cells grown in monoculture versus coculture with human microvascular endothelial cells following multiwalled carbon nanotube exposure. Toxicol Rep 6, 482-488. doi:10.1016/j.toxrep.2019.05.010
Stueckle, T. A., Davidson, D. C., Derk, R. et al. (2018). Short-term pulmonary toxicity assessment of pre- and post-incinerated organomodified nanoclay in mice. ACS Nano 12, 2292-2310. doi:10.1021/acsnano.7b07281
Sun, J., Petersen, E. J., Watson, S. S. et al. (2017). Biophysical characterization of functionalized titania nanoparticles and their application in dental adhesives. Acta Biomater 53, 585-597. doi:10.1016/j.actbio.2017.01.084
Szefler, B. (2018). Nanotechnology, from quantum mechanical calculations up to drug delivery. Int J Nanomedicine 13, 6143-6176. doi:10.2147/IJN.S172907
Tanneberger, K., Knöbel, M., Busser, F. J. M. et al. (2013). Predicting fish acute toxicity using a fish gill cell line-based toxicity assay. Environ Sci Technol 47, 1110-1119. doi:10.1021/es303505z
Taurozzi, J. S., Hackley, V. A. and Wiesner, M. R. (2011). Ultrasonic dispersion of nanoparticles for environmental, health and safety assessment – Issues and recommendations. Nanotoxicology 5, 711-729. doi:10.3109/17435390.2010.528846
Teeguarden, J. G., Hinderliter, P. M., Orr, G. et al. (2007). Particokinetics in vitro: Dosimetry considerations for in vitro nanoparticle toxicity assessments. Toxicol Sci 95, 300-312. doi:10.1093/toxsci/kfl165
Thomas, D. G., Smith, J. N., Thrall, B. D. et al. (2018). ISD3: A particokinetic model for predicting the combined effects of particle sedimentation, diffusion and dissolution on cellular dosimetry for in vitro systems. Part Fibre Toxicol 15, 6. doi:10.1186/s12989-018-0243-7
Uddin, I., Venkatachalam, S., Mukhopadhyay, A. et al. (2016). Nanomaterials in the pharmaceuticals: Occurrence, behaviour and applications. Curr Pharm Des 22, 1472-1484. doi:10.2174/1381612822666160118104727
Wagner, A., White, A. P., Tang, M. C. et al. (2018). Incineration of nanoclay composites leads to byproducts with reduced cellular reactivity. Sci Rep 8, 10709. doi:10.1038/s41598-018-28884-y
Waissi‐Leinonen, G. C., Petersen, E. J., Pakarinen, K. et al. (2012). Toxicity of fullerene (C60) to sediment-dwelling invertebrate Chironomus riparius larvae. Environ Toxicol Chem 31, 2108-2116. doi:10.1002/etc.1926
Wang, L., Castranova, V., Mishra, A. et al. (2010). Dispersion of single-walled carbon nanotubes by a natural lung surfactant for pulmonary in vitro and in vivo toxicity studies. Part Fibre Toxicol 7, 31. doi:10.1186/1743-8977-7-31
Wang, L., Stueckle, T. A., Mishra, A. et al. (2014). Neoplastic-like transformation effect of single-walled and multi-walled carbon nanotubes compared to asbestos on human lung small airway epithelial cells. Nanotoxicology 8, 485-507. doi:10.3109/17435390.2013.801089
Woodruff, R. S., Li, Y., Yan, J. et al. (2012). Genotoxicity evaluation of titanium dioxide nanoparticles using the Ames test and Comet assay. J Appl Toxicol 32, 934-943. doi:10.1002/jat.2781
Wörle-Knirsch, J. M., Pulskamp, K. and Krug, H. F. (2006). Oops they did It again! Carbon nanotubes hoax scientists in viability assays. Nano Lett 6, 1261-1268. doi:10.1021/nl060177c
Xin, X., Barger, M., Roach, K. A. et al. (2020). Toxicity evaluation following pulmonary exposure to an as-manufactured dispersed boron nitride nanotube (BNNT) material in vivo. NanoImpact 19, 100235. doi:10.1016/j.impact.2020.100235
Xu, Z., Liu, Y. and Wang, Y. (2019). Application of Daphnia magna for nanoecotoxicity study. Methods Mol Biol 1894, 345-352. doi:10.1007/978-1-4939-8916-4_21
Youn, S., Wang, R., Gao, J. et al. (2012). Mitigation of the impact of single-walled carbon nanotubes on a freshwater green algae: Pseudokirchneriella subcapitata. Nanotoxicology 6, 161-172. doi:10.3109/17435390.2011.562329
Zhang, M., Xu, C., Jiang, L. et al. (2018). A 3D human lung-on-a-chip model for nanotoxicity testing. Toxicol Res (Camb) 7, 1048-1060. doi:10.1039/c8tx00156a
Zhang, T., Lowry, G. V., Capiro, N. L. et al. (2019). In situ remediation of subsurface contamination: Opportunities and challenges for nanotechnology and advanced materials. Environ Sci Nano 6, 1283-1302. doi:10.1039/C9EN00143C
Zhao, J., Bowman, L., Zhang, X. et al. (2009). Metallic nickel nano- and fine particles induce JB6 cell apoptosis through a caspase-8/AIF mediated cytochrome c-independent pathway. J Nanobiotechnology 7, 2. doi:10.1186/1477-3155-7-2
Zhao, X., Ong, K. J., Ede, J. D. et al. (2013). Evaluating the toxicity of hydroxyapatite nanoparticles in catfish cells and zebrafish embryos. Small 9, 1734-1741. doi:10.1002/smll.201200639
Zhao, Y., Bai, C., Brinker, C. J. et al. (2019). Nano as a rosetta stone: The global roles and opportunities for nanoscience and nanotechnology. ACS Nano 13, 10853-10855. doi:10.1021/acsnano.9b08042