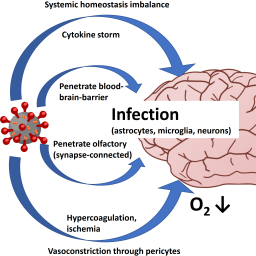
COVID-19 – prime time for microphysiological systems, as illustrated for the brain
Main Article Content
Abstract
The development of therapies for and preventions against infectious diseases depends on the availability of disease models. Bioengineering of human organoids and organs-on-chips is one extremely promising avenue of research. These miniature, laboratory-grown organ systems have been broadly used during the ongoing, unprecedented coronavirus 2019 (COVID-19) pandemic to show the many effects of the etiologic agent, severe acute respiratory coronavirus 2 (SARS-CoV-2) on human organs. In contrast, exposure of most animals either did not result in infection or caused mild clinical signs – not the severe course of the infection suffered by many humans. This article illuminates the opportunities of microphysiological systems (MPS) to study COVID-19 in vitro, with a focus on brain cell infection and its translational relevance to COVID-19 effects on the human brain. Neurovirulence of SARS-CoV-2 has been reproduced in different types of human brain organoids by 10 groups, consistently showing infection of a small portion of brain cells accompanied by limited viral replication. This mirrors increasingly recognized neurological manifestations in COVID-19 patients (evidence of virus infection and brain-specific antibody formation in brain tissue and cerebrospinal fluid). The pathogenesis of neurological signs, their long-term consequences, and possible interventions remain unclear, but future MPS technologies offer prospects to address these open questions.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Abbott, A. (2020). Are infections seeding some cases of Alzheimer’s disease? Nature 587, 22-25. doi:10.1038/d41586-020-03084-9
Abreu, C. M., Gama, L., Krasemann, S. et al. (2018). Microglia increase inflammatory responses in iPSC-dervied human BrainSpheres. Front Microbiol 9, 2766. doi:10.3389/fmicb.2018.02766
Aghagoli, G., Marin, B. G., Katchur, N. J. et al. (2021). Neurological involvement in COVID-19 and potential mechanisms: A review. Neurocrit Care 34, 1062-1071. doi:10.1007/s12028-020-01049-4
Alene, M., Yismaw, L., Assemie, M. A. et al. (2021). Magnitude of asymptomatic COVID-19 cases throughout the course of infection: A systematic review and meta-analysis. PLoS One 16, e0249090. doi:10.1371/journal.pone.0249090
Alexopoulos, H., Magira, E., Bitzogli, K. et al. (2020). Anti-SARS-CoV-2 antibodies in the CSF, blood-brain barrier dysfunction, and neurological outcome: Studies in 8 stuporous and comatose patients. Neurol Neuroimmunol Neuroinflamm 7, e893. doi:10.1212/NXI.0000000000000893
Arbour, N., Day, R., Newcombe, J. et al. (2000). Neuroinvasion by human respiratory coronaviruses. J Virol 74, 8913-8921. doi:10.1128/jvi.74.19.8913-8921.2000.20
Asadi-Pooya, A. A. and Simani, L. (2020). Central nervous system manifestations of COVID-19: A systematic review. J Neurol Sci 413, 116832. doi:10.1016/j.jns.2020.116832
Baig, A., Khaleeq, A., Ali, U. et al. (2020). Evidence of the COVID-19 virus targeting the CNS: Tissue distribution, host-virus interaction, and proposed neurotropic mechanisms. ACS Chem Neurosci 11, 995-998. doi:10.1021/acschemneuro.0c00122
Benameur, K., Agarwal, A., Auld, S. C. et al. (2020). Encephalopathy and encephalitis associated with cerebrospinal fluid cytokine alterations and coronavirus disease. Emerg Infect Dis 26, 2016-2021. doi:10.3201/eid2609.202122
Bodnar, B., Patel, K., Ho, W. et al. (2021). Cellular mechanisms underlying neurological/neuropsychiatric manifestations of COVID-19. J Med Virol 93, 1983-1998. doi:10.1002/jmv.26720
Brann, D. H., Tsukahara, T., Weinreb, C. et al. (2020). Non-neuronal expression of SARS-CoV-2 entry genes in the olfactory system suggests mechanisms underlying COVID-19-associated anosmia. Sci Adv 6, eabc5801. doi:10.1126/sciadv.abc5801
Bullen, C. K., Hogberg, H. T., Bahadirli-Talbott, A. et al. (2020). Infectability of human BrainSphere neurons suggests neurotropism of SARS-CoV-2. ALTEX 37, 665-671. doi:10.14573/altex.2006111
Busquet, F., Hartung, T., Rovida, C. et al. (2020). Harnessing the power of novel animal-free test methods for the development of COVID-19 drugs and vaccines. Arch Toxicol 94, 2263-2272. doi:10.1007/s00204-020-02787-2
Calina, D., Hartung, T., Docea, A. O. et al. (2020). COVID-19 vaccines: Ethical framework concerning human challenge studies. DARU 28, 807-812 doi:10.1007/s40199-020-00371-8
Caporale, N. and Testa, G. (2021). COVID-19 lessons from the dish: Dissecting CNS manifestations through brain organoids. EMBO J 40, e107213. doi:10.15252/embj.2020107213
Chen, K. G., Park, K. and Spence, J. R. (2021). Studying SARS-CoV-2 infectivity and therapeutic responses with complex organoids. Nat Cell Biol 23, 822-833. doi:10.1038/s41556-021-00721-x
Chesnut, M., Hartung, T., Hogberg, H. T. et al. (2021a). Human oligodendrocytes and myelin in vitro to evaluate developmental neurotoxicity. Int J Mol Sci 22, 7929. doi:10.3390/ijms22157929
Chesnut, M., Paschoud, H., Repond, C. et al. (2021b). Human 3D iPSC-derived brain model to study chemical-induced myelin disruption. Int J Mol Sci 22, 9473. doi:10.3390/ijms22179473
Chigr, F., Merzouki, M. and Najimi, M. (2020). Comment on “The neuroinvasive potential of SARS‐CoV‐2 may play a role in the respiratory failure of COVID‐19 patients”. J Med Virol 92, 703-704. doi:10.1002/jmv.25960
Chou, S. H.-Y., Beghi, E., Helbok, R. et al. (2021). Global incidence of neurological manifestations among patients hospitalized with COVID-19 – A report for the GCS-NeuroCOVID consortium and the ENERGY consortium. JAMA Netw Open 4, e2112131. doi:10.1001/jamanetworkopen.2021.12131
Clevers, H. (2020). COVID-19: Organoids go viral. Nat Rev Mol Cell Biol 21, 355-356. doi:10.1038/s41580-020-0258-4
de Melo, B. A. G., Benincasa, J. C., Cruz, E. M. et al. (2021). 3D culture models to study SARS-CoV-2 infectivity and antiviral candidates: From spheroids to bioprinting. Biomed J 44, 31-42. doi:10.1016/j.bj.2020.11.009
Deguchi, S., Serrano-Aroca, A., Tambuwala, M. M. et al. (2021). SARS-CoV-2 research using human pluripotent stem cells and organoids. Stem Cell Transl Med, online ahead of print. doi:10.1002/sctm.21-0183
DeKosky, S. T., Kochanek, P. M., Valadka, A. B. et al. (2021). Blood biomarkers for detection of brain injury in COVID-19 patients. J Neurotrauma 38, 1-43. doi:10.1089/neu.2020.7332
Desforges, M., Le Coupanec, A., Dubeau, P. et al. (2020). Human coronaviruses and other respiratory viruses: Underestimated opportunistic pathogens of the central nervous system? Viruses 12, 14. doi:10.3390/v12010014
Dickinson, P. J. (2020). Coronavirus infection of the central nervous system: Animal models in the time of COVID-19. Front Vet Sci 7, 584673. doi:10.3389/fvets.2020.584673
Dinnon, K. H., Leist, S. R., Schäfer, A. et al. (2020). A mouse-adapted model of SARS-CoV-2 to test COVID-19 countermeasures. Nature 586, 560-566. doi:10.1038/s41586-020-2708-8
Domingues, R. B., Mendes-Correa, M. C., de Moura Leite, F. B. V. et al. (2020). First case of SARS-CoV-2 sequencing in cerebrospinal fluid of a patient with suspected demyelinating disease. J Neurol 267, 3154-3156. doi:10.1007/s00415-020-09996-w
DosSantos, M. F., Devalle, S., Aran, V. et al. (2020). Neuromechanisms of SARS-CoV-2: A review. Front Neuroanat 14, 37. doi:10.3389/fnana.2020.00037
Ellul, M. A., Benjamin, L., Singh, B. et al. (2020). Neurological associations of COVID-19. Lancet Neurol 19, 767-783. doi:10.1016/S1474-4422(20)30221-0
Espíndola, O. M., Brandão, C. O., Gomes, Y. C. al. (2021). Cerebrospinal fluid findings in neurological diseases associated with COVID-19 and insights into mechanisms of disease development. Int J Infect Dis 102, 155-162. doi:10.1016/j.ijid.2020.10.044
Fenizia, C., Biasin, M., Cetin, I. et al. (2020). Analysis of SARS-CoV-2 vertical transmission during pregnancy. Nat Commun 11, 5128. doi:10.1038/s41467-020-18933-4
Finsterer, J. and Scorza, F. A. (2021). Infectious and immune-mediated central nervous system disease in 48 COVID-19 patients. J Clin Neurosci 90, 140-143. doi:10.1016/j.jocn.2021.05.065
Fotuhi, M., Mian, A., Meysami, S. et al. (2020). Neurobiology of COVID-19. J Alzheimers Dis 76, 3-19. doi:10.3233/jad-200581
Franke, C., Ferse, C., Kreye, J. et al. (2020). High frequency of cerebrospinal fluid autoantibodies in COVID-19 patients with neurological symptoms. Brain Behav Immun 93, 415-419. doi:10.1016/j.bbi.2020.12.022
Gasmi, A., Tippairote, T., Mujawdiya, P. K. et al. (2021). Neurological involvements of SARS-CoV2 infection. Mol Neurobiol 58, 944-949. doi:10.1007/s12035-020-02070-6
GBD 2016 Neurology Collaborators (2019). Global, regional, and national burden of neurological disorders, 1990-2016: A systematic analysis for the global burden of disease study 2016. Lancet Neurol 18, 459-480. doi:10.1016/S1474-4422(18)30499-X
Giraudon, P. and Bernard, A. (2010). Inflammation in neuroviral diseases. J Neural Transm 117, 899-906. doi:10.1007/s00702-010-0402-y.18
Gomes, I. C., Karmirian, K., Oliveira, J. et al. (2020). SARS-CoV-2 infection in the central nervous system of a 1-year-old infant submitted to complete autopsy. Preprints. doi:10.20944/preprints202009.0297.v1
Graham, M. L. and Prescott, M. J. (2015). The multifactorial role of the 3Rs in shifting the harm-benefit analysis in animal models of disease. Eur J Pharmacol 759, 19-29. doi:10.1016/j.ejphar.2015.03.040
Granja, M. G., Oliveira, A. C. R., de Figueiredo, C. S. et al. (2021). SARS-CoV-2 infection in pregnant women: Neuroimmune-endocrine changes at the maternal-fetal interface. Neuroimmunomodulation 28, 1-21. doi:10.1159/000515556
Harschnitz, O. and Studer, L. (2021). Human stem cell models to study host-virus interactions in the central nervous system. Nat Rev Immunol 21, 441-453. doi:10.1038/s41577-020-00474-y
Hartung, T. (2010). Comparative analysis of the revised Directive 2010/63/EU for the protection of laboratory animals with its predecessor 86/609/EEC – A t4 report. ALTEX 27, 285-303. doi:10.14573/altex.2010.4.285
Hartung, T. and Zurlo, J. (2012). Alternative approaches for medical countermeasures to biological and chemical terrorism and warfare. ALTEX 29, 251-260. doi:10.14573/altex.2012.3.251
Hartung, T. (2013). Look back in anger – What clinical studies tell us about preclinical work. ALTEX 30, 275-291. doi:10.14573/altex.2013.3.275
Hascup, E. R. and Hascup, K. N. (2020). Does SARS-CoV-2 infection cause chronic neurological complications? Geroscience 42, 1083-1087. doi:10.1007/s11357-020-00207-y
Hogberg, H. T., de Cássia da Silveira e Sá, R., Kleensang, A. et al. (2021). Organophosphorus flame retardants are developmental neurotoxicants in a rat primary BrainSphere in vitro model. Arch Toxicol 95, 207-228, doi:10.1007/s00204-020-02903-2
Imai, M., Iwatsuki-Horimoto, K., Hatta, M. et al. (2020). Syrian hamsters as a small animal model for SARS-CoV-2 infection and countermeasure development. Proc Natl Acad Sci U S A 117, 16587-16595. doi:10.1073/pnas.2009799117
Jacob, F., Pather, S. R., Huang, W.-K. et al. (2020). Human pluripotent stem cell-derived neural cells and brain organoids reveal SARS-CoV-2 neurotropism predominates in choroid plexus epithelium. Cell Stem Cell 27, 937-950.e9. doi:10.1016/j.stem.2020.09.016
Kanberg, N., Ashton, N. J., Andersson, L.-M. et al. (2020). Neurochemical evidence of astrocytic and neuronal injury commonly found in COVID-19. Neurology 95, e1754-e1759. doi:10.1212/WNL.0000000000010111
Keddie, S., Pakpoor, J., Mousele, C. et al. (2021). Epidemiological and cohort study finds no association between COVID-19 and Guillain-Barré syndrome. Brain 144, 682-693. doi:10.1093/brain/awaa433
Kleinstreuer, N. and Holmes, A. (2021). Harnessing the power of microphysiological systems for COVID-19 research. Drug Discov Today, online ahead of print. doi:10.1016/j.drudis.2021.06.020
Komine-Aizawa, S., Takada, K. and Hayakawa, S. (2020). Placental barrier against COVID-19. Placenta 99, 45-49. doi:10.1016/j.placenta.2020.07.022
Koralnik, I. J. and Tyler, K. L. (2020). COVID-19: A global threat to the nervous system. Ann Neurol 88, 1-11. doi:10.1002/ana.25807
Lau, K. K., Yu, W. C., Chu, C. M. et al. (2004). Possible central nervous system infection by SARS coronavirus. Emerg Infect Dis 10, 342-344. doi:10.3201/eid1002.030638.22
Li, H., Xue, Q. and Xu, X. (2020). Involvement of the nervous system in SARS-CoV-2 infection. Neurotox Res 38, 1-7. doi:10.1007/s12640-020-00219-8
Li, Y.-C., Zhang, Y. and Tan, B.-H. (2021). What can cerebrospinal fluid testing and brain autopsies tell us about viral neuroinvasion of SARS-CoV-2. J Med Virol 93, 4247-4257. doi:10.1002/jmv.26943
Lima, M., Siokas, V., Aloizou, A.-M. et al. (2020). Unraveling the possible routes of SARS-COV-2 invasion into the central nervous system. Curr Treat Options Neurol 22, 37. doi:10.1007/s11940-020-00647-z
Lins, B. (2021). Maternal immune activation as a risk factor for psychiatric illness in the context of the SARS-CoV-2 pandemic. Brain Behav Immun Health 16, 100297. doi:10.1016/j.bbih.2021.100297
Liu, J.-M., Tan, B.-H., Wu, S. et al. (2021). Evidence of central nervous system infection and neuroinvasive routes, as well as neurological involvement, in the lethality of SARS-CoV-2 infection. J Med Virol 93, 1304-1313. doi:10.1002/jmv.26570
Lopez-Leon, S., Wegman-Ostrosky, T., Perelman, C. et al. (2021). More than 50 long-term effects of COVID-19: A systematic review and meta-analysis. Sci Rep 11, 16144. doi:10.1038/s41598-021-95565-8
Lou, J. J., Movassaghi, M., Gordy, D. et al. (2021). Neuropathology of COVID-19 (neuro-COVID): Clinicopathological update. Free Neuropathol 2, 2. doi:10.17879/freeneuropathology-2021-2993
Luis, M. B., Liguori, N. F., López, P. A. et al. (2021). SARS-CoV-2 RNA detection in cerebrospinal fluid: Presentation of two cases and review of literature. Brain Behav Immun Health 15, 100282. doi:10.1016/j.bbih.2021.100282
Mahalakshmi, A. M., Ray, B., Tuladhar, S. et al. (2021). Does COVID-19 contribute to development of neurological disease? Immun Inflamm Dis 9, 48-58. doi:10.1002/iid3.387
Mallapaty, S. (2020). Mini organs reveal how the coronavirus ravages the body. Nature 583, 15-16. doi:10.1038/d41586-020-01864-x
Mao, L., Jin, H., Wang, M. et al. (2020). Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurology 77, 683-690. doi:10.1001/jamaneurol.2020.1127
Mao, X.-Y. and Jin, W.-L. (2020). iPSCs-derived platform: A feasible tool for probing the neurotropism of SARS-CoV‑2. ACS Chem Neurosci 11, 2489-2491. doi:10.1021/acschemneuro.0c00512
Marinho, P. S., da Cunha, A. J. L. A., Chimelli, L. et al. (2021). Case report: SARS-CoV-2 mother-to-child transmission and fetal death associated with severe placental thromboembolism. Front Med 8, 677001. doi:10.3389/fmed.2021.677001
Marra, D. E., Busl, K. M. and Robinson, C. P (2021). Examination of early CNS symptoms and severe coronavirus disease 2019: A multicenter observational case series. Crit Care Explor 3, e0456. doi:10.1097/CCE.0000000000000456
Marshall, M. (2021). COVID and the brain: Researchers zero in on how damage occurs. Nature 595, 484-485. doi:10.1038/d41586-021-01693-6
Marx, U., Andersson, T. B., Bahinski, A. et al. (2016). Biology-inspired microphysiological system approaches to solve the prediction dilemma of substance testing using animals. ALTEX 33, 272-321. doi:10.14573/altex.1603161
Marx, U., Akabane, T., Andersson, T. B. et al. (2020). Biology-inspired microphysiological systems to advance medicines for patient benefit and animal welfare. ALTEX 37, 365-394. doi:10.14573/altex.2001241
Matschke, J., Lütgehetmann, M., Hagel, C. et al. (2020). Neuropathology of patients with COVID-19 in Germany: A post-mortem case series. Lancet Neurol 19, 919-929. doi:10.1016/S1474-4422(20)30308-2
Maury, A., Lyoubi, A., Peiffer-Smadja, N. et al. (2021). Neurological manifestations associated with SARS-CoV-2 and other coronaviruses: A narrative review for clinicians. Rev Neurol 177, 51-64. doi:10.1016/j.neurol.2020.10.001
McMahon, C. L., Staples, H., Gazi, M. et al. (2021). SARS-CoV-2 targets glial cells in human cortical organoids. Stem Cell Reports 16, 1156-1164. doi:10.1016/j.stemcr.2021.01.016
Meigs, L., Smirnova, L., Rovida, C. et al. (2018). Animal testing and its alternatives – the most important omics is economics. ALTEX 35, 275-305. doi:10.14573/altex.1807041
Meinhardt, J., Radke, J., Dittmayer, C. et al. (2021). Olfactory transmucosal SARS-CoV-2 invasion as a port of central nervous system entry in individuals with COVID-19. Nat Neurosci 24, 168-175. doi:10.1038/s41593-020-00758-5
Miners, S., Kehoe, P. G. and Love, S. (2020). Cognitive impact of COVID-19: Looking beyond the short term. Alzheimers Res Ther 12, 170. doi:10.1186/s13195-020-00744-w
Modafferi, S., Zhong, X., Kleensang, A. et al. (2021). Gene-environment interactions in developmental neurotoxicity: A case study of synergy between chlorpyrifos and CHD8 knockout in human BrainSpheres. Environ Health Perspect 129, 77001. doi:10.1289/EHP8580
Monchatre-Leroy, E., Lesellier, S. and Wasniewski, M. (2021). Hamster and ferret experimental infection with intranasal low dose of a single strain of SARS-CoV-2. J Gen Virol 102. doi:10.1099/jgv.0.001567
Montalvan, V., Lee, J., Bueso, T. et al. (2020). Neurological manifestations of Covid-19 and other coronavirus infections: A systematic review. Clin Neurol Neurosurg 194, 105921. doi:101016/j.clineuro.2020.105921
Moriguchi, T., Harii, N., Goto, J. et al. (2020). A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int J Infect Dis 94, 55-58. doi:10.1016/j.ijid.2020.03.062
Muñoz-Fontela, C., Dowling, W. E., Funnell, S. G. P. et al. (2020). Animal models for COVID-19. Nature 586, 509-515. doi:10.1038/s41586-020-2787-6
Murta, V., Villarreal, A. and Ramos, A. J. (2020). Severe acute respiratory syndrome coronavirus 2 impact on the central nervous system: Are astrocytes and microglia main players or merely bystanders? ASN Neuro 12, 1759091420954960. doi:10.1177/1759091420954960
Najjar, S., Najjar, A., Chong, D. J. et al. (2020). Central nervous system complications associated with SARS-CoV-2 infection: Integrative concepts of pathophysiology and case reports. J Neuroinflammation 17, 231. doi:10.1186/s12974-020-01896-0
Najt, P., Richards, H. L. and Fortune, D. G. (2021), Brain imaging in patients with COVID-19: A systematic review. Brain Behav Immun Health 16, 100290. doi:10.1016/j.bbih.2021.100290
Nathanson, N. and Langmuir, A. D. (1963). The Cutter incident. Poliomyelitis following formaldehyde-inactivated poliovirus vaccination in the United States during the Spring of 1955. II. Relationship of poliomyelitis to Cutter vaccine. 1963. Am J Hyg 78, 29-60. doi:10.1093/oxfordjournals.aje.a120328
Natoli, S., Oliveira, V., Calabresi, P. et al. (2020). Does SARS‐Cov‐2 invade the brain? Translational lessons from animal models. Eur J Neurol 27, 1764-1773. doi:10.1111/ene.14277
Ng, J. H., Sun, A., Je, H. S. et al. (2021). Unravelling pathophysiology of neurological and psychiatric complications of COVID-19 using brain organoids. Neuroscientist, 10738584211015136. doi:10.1177/10738584211015136
Nymark, P., Sachana, M., Leite, S. B. et al. (2021). Systematic organization of COVID-19 data supported by the adverse outcome pathway framework. Front Public Health 9, 638605. doi:10.3389/fpubh.2021.638605
Pallanti, S., Grassi, E., Makris, N. et al. (2020). Neurocovid-19: A clinical neuroscience-based approach to reduce SARS-CoV-2 related mental health sequelae. J Psychiatr Res 130, 215-217. doi:10.1016/j.jpsychires.2020.08.008
Pamies, D., Barreras, P., Block, K. et al. (2017). A human brain microphysiological system derived from iPSC to study central nervous system toxicity and disease. ALTEX 34, 362-376. doi:10.14573/altex.1609122
Pamies, D., Block, K., Lau, P. al. (2018). Rotenone exerts developmental neurotoxicity in a human brain spheroid model. Toxicol Appl Pharmacol 354, 101-114. doi:10.1016/j.taap.2018.02.003
Pamies, D., Leist, M., Coecke, S. et al. (2020). Good cell and tissue culture practice 2.0 (GCCP 2.0) – Draft for stakeholder discussion and call for action. ALTEX 37, 490-492. doi:10.14573/altex.2007091
Park, S. E., Georgescu, A. and Huh, D. (2019). Organoids-on-a-chip. Science 364, 960-965. doi:10.1126/science.aaw7894
Pedrosa, C. D. S. G., Goto-Silva, L., Temerozo, J. R. et al. (2021). Non-permissive SARS-CoV-2 infection in human neurospheres. Stem Cell Res 54, 102436. doi:10.1016/j.scr.2021.102436
Pellegrini, L., Albecka, A., Mallery, D. L. et al. (2020). SARS-CoV-2 infects the brain choroid plexus and disrupts the blood-CSF barrier in human brain organoids. Cell Stem Cell 27, 951-961.e5. doi:10.1016/j.stem.2020.10.001
Perlman, S. and Netland, J. (2009). Corona viruses post-SARS: Update on replication and pathogenesis. Nat Rev Microbiol 7, 439-450. doi:10.1038/nrmicro2147.21
Pezzini, A. and Padovani, A. (2020). Lifting the mask on neurological manifestations of COVID-19. Nat Rev Neurol 16, 636-644. doi:10.1038/s41582-020-0398-3
Pouga, L. (2021). Encephalitic syndrome and anosmia in COVID-19: Do these clinical presentations really reflect SARS-CoV-2 neurotropism? A theory based on the review of 25 COVID-19 cases. J Med Virol 93, 550-558. doi:10.1002/jmv.26309
Poyiadji, N., Shahin, G., Noujaim, D. et al. (2020). COVID-19-associated acute hemorrhagic necrotizing encephalopathy: Imaging features. Radiology 296, E119-E120. doi:10.1148/radiol.2020201187
Pronker, E. S., Weenen, T. C., Commandeur, H. et al. (2013). Risk in vaccine research and development quantified. PLoS One 8, e57755. doi:10.1371/journal.pone.0057755
Ramani, A., Müller, L., Ostermann, P. N. et al. (2020). SARS-CoV-2 targets neurons of 3D human brain organoids. EMBO J 39, e106230. doi:10.15252/embj.2020106230
Ramani, A., Pranty, A. I. and Gopalakrishnan, J. (2021). Neurotropic effects of SARS-CoV-2 modeled by the human brain organoids. Stem Cell Reports 16, 373-384. doi:10.1016/j.stemcr.2021.02.007
Ray, S. T. J., Abdel-Mannan, O., Sa, M. et al. (2021). Neurological manifestations of SARS-CoV-2 infection in hospitalised children and adolescents in the UK: A prospective national cohort study. Lancet Child Adolesc Health 5, 631-641. doi:10.1016/S2352-4642(21)00193-0
Rogers, J. P., Watson, C. J., Badenoch, J. et al. (2021). Neurology and neuropsychiatry of COVID-19: A systematic review and meta-analysis of the early literature reveals frequent CNS manifestations and key emerging narratives. J Neurol Neurosurg Psychiatry 92, 932-941. doi:10.1136/jnnp-2021-326405
Roth, A. and MPS-WS Berlin 2019 (2021). Human microphysiological systems for drug development. Science 373, 1304-1306. doi:10.1126/science.abc3734
Sanclemente-Alaman, I., Moreno-Jiménez, L., Benito-Martín, M. S. et al. (2020). Experimental models for the study of central nervous system infection by SARS-CoV-2. Front Immunol 11, 2163. doi:10.3389/fimmu.2020.02163
Shan, C., Yao, Y. F., Yang, X. L. et al. (2020). Infection with novel coronavirus (SARS-CoV-2) causes pneumonia in Rhesus macaques. Cell Res 30, 670-677. doi:10.1038/s41422-020-0364-z
Sheraton, M., Deo, N., Kashyap, R. et al. (2020). A review of neurological complications of COVID-19. Cureus 12, e8192. doi:10.7759/cureus.8192
Shuid, A. N., Jayusman, P. A., Shuid, N. et al. (2021). Association between viral infections and risk of autistic disorder: An overview. Int J Environ Res Public Health 18, 2817. doi:10.3390/ijerph18062817
Solomon, I. H. and Normandin, E. (2020). Neuropathological features of Covid-19. N Engl J Med 383, 989-992. doi:10.1056/NEJMc2019373
Song, E., Zhang, C., Israelow, B. et al. (2021a). Neuroinvasion of SARS-CoV-2 in human and mouse brain. J Exp Med 218, e20202135. doi:10.1084/jem.20202135
Song, E., Bartley, C. M., Chow, R. D. et al. (2021b). Divergent and self-reactive immune responses in the CNS of COVID-19 patients with neurological symptoms. Cell Rep Med 2, 100288. doi:10.1016/j.xcrm.2021.100288
Steardo, L. Jr., Steardo, L. and Verkhratsky, A. (2020). Psychiatric face of COVID-19. Transl Psychiatry 10, 261. doi:10.1038/s41398-020-00949-5
Stevens, R. (2020). How Does Coronavirus Affect the Brain? Johns Hopkins Medicine. https://www.hopkinsmedicine.org/health/conditions-and-diseases/coronavirus/how-does-coronavirus-affect-the-brain
Szcześniak, D., Gładka, A., Misiak, B. et al. (2021). The SARS-CoV-2 and mental health: From biological mechanisms to social consequences. Prog Neuropsychopharmacol Biol Psychiatry 104, 110046. doi:10.1016/j.pnpbp.2020.110046
Taglauer, E., Benarroch, Y., Rop, K. et al. (2020). Consistent localization of SARS-CoV-2 spike glycoprotein and ACE2 over TMPRSS2 predominance in placental villi of 15 COVID-19 positive maternal-fetal dyads. Placenta 100, 69-74. doi:10.1016/j.placenta.2020.08.015
Tang, H., Abouleila, Y., Si, L. et al. (2020). Human organs-on-chips for virology. Trends Microbiol 28, 934-946. doi:10.1016/j.tim.2020.06.005
Tiwari, S. K., Wang, S., Smith, D. et al. (2021). Revealing tissue-specific SARS-CoV-2 infection and host responses using human stem cell derived lung and cerebral organoids. Stem Cell Rep 16, 437-445. doi:10.1016/j.stemcr.2021.02.005
Troyer, E. A., Kohn, J. N. and Hong, S. (2020). Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain Behav Immun 87, 34-39. doi:10.1016/j.bbi.2020.04.027
Valiuddin, H. M., Kalajdzic, A., Rosati, J. et al. (2020). Update on neurological manifestations of SARS-CoV-2. West J Emerg Med 21, 45-51. doi:10.5811/westjem.2020.8.48839
Vitalakumar, D., Sharma, A., Kumar, A. et al. (2021). Neurological manifestations in COVID-19 patients: A meta-analysis. ACS Chem Neurosci 15, 2776-2797. doi:10.1021/acschemneuro.1c00353
Wang, C., Zhang, M., Garcia, G. Jr. et al. (2021). ApoE-isoform-dependent SARS-CoV-2 neurotropism and cellular response. Cell Stem Cell 28, 331-342.e5. doi:10.1016/j.stem.2020.12.018
Wang, L., Sievert, D., Clark, A. E. et al. (2021). A human three-dimensional neural-perivascular ‘assembloid’ promotes astrocytic development and enables modeling of SARS-CoV-2 neuropathology. Nat Med 27, 1600-1606. doi:10.1038/s41591-021-01443-1
Willner, M. J., Xiao, Y., Kim, H. S. et al. (2021). Modeling SARS-CoV-2 infection in individuals with opioid use disorder with brain organoids. J Tissue Eng 12, 2041731420985299. doi:10.1177/2041731420985299
Wittwehr, C., Amorim, M. J., Clerbaux, L.-A. et al. (2021). Understanding COVID-19 through adverse outcome pathways – 2nd CIAO AOP Design Workshop. ALTEX 38, 351-357. doi:10.14573/altex.2102221
Wu, Y., Xu, X., Chen, Z. et al. (2020). Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav Immun 87, 18-22. doi:10.1016/j.bbi.2020.03.031
Yachou, Y., El Idrissi, A., Belapasov, V. et al. (2020). Neuroinvasion, neurotropic, and neuroinflammatory events of SARS-CoV-2: Understanding the neurological manifestations in COVID-19 patients. Neurol Sci 41, 2657-2669. doi:10.1007/s10072-020-04575-3
Yi, S. A., Nam, K. H., Yun, J. et al. (2020). Infection of brain organoids and 2D cortical neurons with SARS-CoV-2 pseudovirus. Viruses 12, 1004. doi:10.3390/v12091004
Yinda, C. K., Port, J. R., Bushmaker, T. et al. (2021). K18-hACE2 mice develop respiratory disease resembling severe COVID-19. PLoS Pathog 17, e1009195. doi:10.1371/journal.ppat.1009195
Zanin, L., Saraceno, G., Panciani, P. P. et al. (2020). SARS-CoV-2 can induce brain and spine demyelinating lesions. Acta Neurochir 162, 1491-1494. doi:10.1007/s00701-020-04374-x
Zhang, B. Z., Chu, H., Han, S. et al. (2020). SARS-CoV-2 infects human neural progenitor cells and brain organoids. Cell Res 30, 928-931. doi:10.1038/s41422-020-0390-x
Zhang, L., Zhou, L., Bao, L. et al. (2021). SARS-CoV-2 crosses the blood-brain barrier accompanied with basement membrane disruption without tight junctions alteration. Signal Transduct Target Ther 6, 337. doi:10.1038/s41392-021-00719-9
Zhong, X., Harris, G., Smirnova, L. et al. (2020). Paroxetine exerts developmental neurotoxicity in an iPSC derived 3D human brain model. Front Cell Neurosci 14, 25. doi:10.3389/fncel.2020.00025
Zhou, Z., Kang, H., Li, S. et al. (2020). Understanding the neurotropic characteristics of SARS-CoV-2: From neurological manifestations of COVID-19 to potential neurotropic mechanisms. J Neurol 267, 2179-2184. doi:10.1007/s00415-020-09929-7