A novel coculture system for assessing respiratory sensitizing potential by IL-4 in T cells
Main Article Content
Abstract
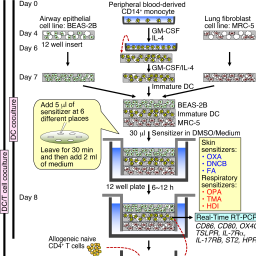
Although several in vitro assays that predict the sensitizing potential of chemicals have been developed, none can distinguish between chemical respiratory and skin sensitizers. Recently, we established a new three-dimensional dendritic cell (DC) coculture system consisting of a human airway epithelial cell line, immature DCs derived from human peripheral monocytes, and a human lung fibroblast cell line. In this coculture system, compared to skin sensitizers, respiratory sensitizers showed enhanced mRNA expression in DCs of the key costimulatory molecule OX40 ligand (OX40L), which is important for T helper 2 (Th2) cell differentiation. Herein, we established a new two-step DC/T cell coculture system by adding peripheral allogeneic naïve CD4+ T cells to the DCs stimulated in the DC coculture system. In this DC/T cell coculture system, model respiratory sensitizers, but not skin sensitizers, enhanced mRNA expression of the predominant Th2 marker interleukin-4 (IL-4). To improve the versatility, in place of peripheral monocytes, monocyte-derived proliferating cells called CD14-ML were used in the DC coculture system. As in peripheral monocytes, enhanced mRNA expression of OX40L was induced in CD14-ML by respiratory sensitizers compared to skin sensitizers. When these cell lines were applied to the DC/T cell coculture system with peripheral allogeneic naïve CD4+ T cells, respiratory sensitizers but not skin sensitizers enhanced the mRNA expression of IL-4. Thus, this DC/T cell coculture system may be useful for discriminating between respiratory and skin sensitizers by differential mRNA upregulation of IL-4 in T cells.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Adenuga, D., Woolhiser, M. R., Gollapudi, B. B. et al. (2012). Differential gene expression responses distinguish contact and respiratory sensitizers and nonsensitizing irritants in the local lymph node assay. Toxicol Sci 126, 413-425. doi:10.1093/toxsci/kfs071
Arts, J. H., de Jong, W. H., van Triel, J. J. et al. (2008). The respiratory local lymph node assay as a tool to study respiratory sensitizers. Toxicol Sci 106, 423-434. doi:10.1093/toxsci/kfn199
Arts, J. (2020). How to assess respiratory sensitization of low molecular weight chemicals? Int J Hyg Environ Health 225, 113469. doi:10.1016/j.ijheh.2020.113469 Ashikaga, T., Yoshida, Y., Hirota, M. et al. (2006). Development of an in vitro skin sensitization test using human cell lines: the human Cell Line Activation Test (h-CLAT). I. Optimization of the h-CLAT protocol. Toxicol In Vitro 20, 767-773. doi:10.1016/j.tiv.2005.10.012
Balharry, D., Sexton, K., BeruBe, K. A. (2008). An in vitro approach to assess the toxicity of inhaled tobacco smoke components: nicotine, cadmium, formaldehyde and urethane. Toxicology 244, 66-76. doi:10.1016/j.tox.2007.11.001
Banchereau, J., Steinman, R. M. (1998). Dendritic cells and the control of immunity. Nature 392, 245-252. doi:10.1038/32588
Bendfeldt, H., Benary, M., Scheel, T. et al. (2012). Stable IL-2 decision making by endogenous c-Fos amounts in peripheral memory T-helper cells. J Biol Chem 287, 18386-18397. doi:10.1074/jbc.M112.358853
Chackerian, A. A., Oldham, E. R., Murphy, E. E. et al. (2007). IL-1 receptor accessory protein and ST2 comprise the IL-33 receptor complex. J Immunol 179, 2551-2555. doi:10.4049/jimmunol.179.4.2551
Chary, A., Hennen, J., Klein, S. G. et al. (2018). Respiratory sensitization: toxicological point of view on the available assays. Arch Toxicol 92, 803-822. doi:10.1007/s00204-017-2088-5
Chipinda, I., Hettick, J. M., Siegel, P. D. (2011). Haptenation: chemical reactivity and protein binding. J Allergy (Cairo) 2011, 839682. doi:10.1155/2011/839682
De Jong, W. H., Arts, J. H., De Klerk, A. et al. (2009). Contact and respiratory sensitizers can be identified by cytokine profiles following inhalation exposure. Toxicology 261, 103-111. doi:10.1016/j.tox.2009.04.057
Dearman, R. J., Basketter, D. A., Kimber, I. (1995). Differential cytokine production following chronic exposure of mice to chemical respiratory and contact allergens. Immunology 86, 545-550.
Dearman, R. J., Betts, C. J., Caddick, H. T. et al. (2008). Cytokine profiling of chemical allergens in mice: measurement of message versus protein. Toxicology 252, 17-25. doi:10.1016/j.tox.2008.07.055
Furue, M., Furue, M. (2021). OX40L-OX40 Signaling in Atopic Dermatitis. J Clin Med 10. doi:10.3390/jcm10122578
Gerberick, G. F., Vassallo, J. D., Bailey, R. E. et al. (2004). Development of a peptide reactivity assay for screening contact allergens. Toxicol Sci 81, 332-343. doi:10.1093/toxsci/kfh213
Goutet, M., Pepin, E., Langonne, I. et al. (2012). Identification of contact and respiratory sensitizers according to IL-4 receptor alpha expression and IL-2 production. Toxicol Appl Pharmacol 260, 95-104. doi:10.1016/j.taap.2012.02.009
Haruta, M., Tomita, Y., Imamura, Y. et al. (2013). Generation of a large number of functional dendritic cells from human monocytes expanded by forced expression of cMYC plus BMI1. Hum Immunol 74, 1400-1408. doi:10.1016/j.humimm.2013.05.017
Hou, F., Xing, C., Li, B. et al. (2020). Performance of a novel in vitro assay for skin sensitization based on activation of T lymphocytes. ALTEX 37, 451-468. doi:10.14573/altex.2001312
Huang, S., Wiszniewski, L., Constant, S. et al. (2013). Potential of in vitro reconstituted 3D human airway epithelia (MucilAir) to assess respiratory sensitizers. Toxicol In Vitro 27, 1151-1156. doi:10.1016/j.tiv.2012.10.010
Imamura, Y., Haruta, M., Tomita, Y. et al. (2016). Generation of large numbers of antigen-expressing human dendritic cells using CD14-ML technology. PLoS One 11, e0152384. doi:10.1371/journal.pone.0152384
Ito, T., Wang, Y. H., Duramad, O. et al. (2005). TSLP-activated dendritic cells induce an inflammatory T helper type 2 cell response through OX40 ligand. J Exp Med 202, 1213-1223. doi:10.1084/jem.20051135
Jacobs, J. P., Jones, C. M., Baille, J. P. (1970). Characteristics of a human diploid cell designated MRC-5. Nature 227, 168-170. doi:10.1038/227168a0
Kimber, I., Dearman, R. J., Scholes, E. W. et al. (1994). The local lymph node assay: developments and applications. Toxicology 93, 13-31. doi:10.1016/0300-483x(94)90193-7
Kimber, I., Poole, A., Basketter, D. A. (2018). Skin and respiratory chemical allergy: confluence and divergence in a hybrid adverse outcome pathway. Toxicol Res (Camb) 7, 586-605. doi:10.1039/c7tx00272f
Kimura, Y., Fujimura, C., Ito, Y. et al. (2015). Optimization of the IL-8 Luc assay as an in vitro test for skin sensitization. Toxicol In Vitro 29, 1816-1830. doi:10.1016/j.tiv.2015.07.006
Knight, E., Murray, B., Carnachan, R. et al. (2011). Alvetex(R): polystyrene scaffold technology for routine three dimensional cell culture. Methods Mol Biol 695, 323-340. doi:10.1007/978-1-60761-984-0_20
Lakkis, F. G., Lechler, R. I. (2013). Origin and biology of the allogeneic response. Cold Spring Harb Perspect Med 3, a012933.. doi:10.1101/cshperspect.a014993
MacKay, C., Davies, M., Summerfield, V. et al. (2013). From pathways to people: applying the adverse outcome pathway (AOP) for skin sensitization to risk assessment. ALTEX 30, 473-486. doi:10.14573/altex.2013.4.473
Mizoguchi, I., Ohashi, M., Chiba, Y. et al. (2017). Prediction of chemical respiratory and contact sensitizers by OX40L expression in dendritic cells using a novel 3D coculture system. Front Immunol 8, 929. doi:10.3389/fimmu.2017.00929
Natsch, A., Emter, R. (2016). Nrf2 activation as a key event triggered by skin sensitisers: The development of the stable KeratinoSens reporter gene assay. Altern Lab Anim 44, 443-451. doi:10.1177/026119291604400513
North, C. M., Ezendam, J., Hotchkiss, J. A. et al. (2016). Developing a framework for assessing chemical respiratory sensitization: A workshop report. Regul Toxicol Pharmacol 80, 295-309. doi:10.1016/j.yrtph.2016.06.006
Pandey, A., Ozaki, K., Baumann, H. et al. (2000). Cloning of a receptor subunit required for signaling by thymic stromal lymphopoietin. Nat Immunol 1, 59-64. doi:10.1038/76923
Parkinson, E., Aleksic, M., Cubberley, R. et al. (2018). Determination of protein haptenation by chemical sensitizers within the complexity of the human skin proteome. Toxicol Sci 162, 429-438. doi:10.1093/toxsci/kfx265
Paul, W. E. (2015). History of interleukin-4. Cytokine 75, 3-7. doi:10.1016/j.cyto.2015.01.038
Reddel, R. R., Ke, Y., Gerwin, B. I. et al. (1988). Transformation of human bronchial epithelial cells by infection with SV40 or adenovirus-12 SV40 hybrid virus, or transfection via strontium phosphate coprecipitation with a plasmid containing SV40 early region genes. Cancer Res 48, 1904-1909.
Richter, A., Schmucker, S. S., Esser, P. R. et al. (2013). Human T cell priming assay (hTCPA) for the identification of contact allergens based on naive T cells and DC IFN-gamma and TNF-alpha readout. Toxicol In Vitro 27, 1180-1185. doi:10.1016/j.tiv.2012.08.007
Rickel, E. A., Siegel, L. A., Yoon, B. R. et al. (2008). Identification of functional roles for both IL-17RB and IL-17RA in mediating IL-25-induced activities. J Immunol 181, 4299-4310. doi:10.4049/jimmunol.181.6.4299
Roan, F., Obata-Ninomiya, K., Ziegler, S. F. (2019). Epithelial cell-derived cytokines: more than just signaling the alarm. J Clin Invest 129, 1441-1451. doi:10.1172/JCI124606
Roggen, E. L. (2014). In vitro approaches for detection of chemical sensitization. Basic Clin Pharmacol Toxicol 115, 32-40. doi:10.1111/bcpt.12202
Sadekar, N., Boisleve, F., Dekant, W. et al. (2021). Identifying a reference list of respiratory sensitizers for the evaluation of novel approaches to study respiratory sensitization. Crit Rev Toxicol 51, 792-804. doi:10.1080/10408444.2021.2024142
Sherman, L. A., Chattopadhyay, S. (1993). The molecular basis of allorecognition. Annu Rev Immunol 11, 385-402. doi:10.1146/annurev.iy.11.040193.002125
Sullivan, K. M., Enoch, S. J., Ezendam, J. et al. (2017). An adverse outcome pathway for sensitization of the respiratory tract by low-molecular-weight chemicals: building evidence to support the utility of in vitro and in silico methods in a regulatory context. Appl Vitro Toxicol 3, 213-226. doi:org/10.1089/aivt.2017.0010
Szabo, S. J., Kim, S. T., Costa, G. L. et al. (2000). A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell 100, 655-669. doi:10.1016/s0092-8674(00)80702-3
Tarlo, S. M., Lemiere, C. (2014). Occupational asthma. N Engl J Med 370, 640-649. doi:10.1056/NEJMra1301758
Tha, E. L., Canavez, A., Schuck, D. C. et al. (2021). Beyond dermal exposure: The respiratory tract as a target organ in hazard assessments of cosmetic ingredients. Regul Toxicol Pharmacol 124, 104976. doi:10.1016/j.yrtph.2021.104976
van Vliet, E., Kuhnl, J., Goebel, C. et al. (2018). State-of-the-art and new options to assess T cell activation by skin sensitizers: Cosmetics Europe Workshop. ALTEX 35, 179-192. doi:10.14573/altex.1709011
Vandebriel, R. J., De Jong, W. H., Spiekstra, S. W. et al. (2000). Assessment of preferential T-helper 1 or T-helper 2 induction by low molecular weight compounds using the local lymph node assay in conjunction with RT-PCR and ELISA for interferon-gamma and interleukin-4. Toxicol Appl Pharmacol 162, 77-85. doi:10.1006/taap.1999.8841
Vandebriel, R. J., Gremmer, E. R., van Hartskamp, M. et al. (2007). Effects of a diphtheria-tetanus-acellular pertussis vaccine on immune responses in murine local lymph node and lung allergy models. Clin Vaccine Immunol 14, 211-219. doi:10.1128/CVI.00306-06
Vanoirbeek, J. A., Tarkowski, M., Vanhooren, H. M. et al. (2006). Validation of a mouse model of chemical-induced asthma using trimellitic anhydride, a respiratory sensitizer, and dinitrochlorobenzene, a dermal sensitizer. J Allergy Clin Immunol 117, 1090-1097. doi:10.1016/j.jaci.2006.01.027
Vocanson, M., Achachi, A., Mutez, V. et al. (2014). Human T cell priming assay: depletion of peripheral blood lymphocytes in CD25+ cells improves the in vitro detection of weak allergen-specific T cells. Exp Suppl 104, 89-100. doi:10.1007/978-3-0348-0726-5_7
Weninger, W., von Andrian, U. H. (2003). Chemokine regulation of naive T cell traffic in health and disease. Semin Immunol 15, 257-270. doi:10.1016/j.smim.2003.08.007
Zhou, M., Ouyang, W. (2003). The function role of GATA-3 in Th1 and Th2 differentiation. Immunol Res 28, 25-37. doi:10.1385/IR:28:1:25
Zhu, J. (2015). T helper 2 (Th2) cell differentiation, type 2 innate lymphoid cell (ILC2) development and regulation of interleukin-4 (IL-4) and IL-13 production. Cytokine 75, 14-24. doi:10.1016/j.cyto.2015.05.010
Ziegler, S. F., Roan, F., Bell, B. D. et al. (2013). The biology of thymic stromal lymphopoietin (TSLP). Adv Pharmacol 66, 129-155. doi:10.1016/B978-0-12-404717-4.00004-4