Open-source human skin model with an in vivo-like barrier for drug testing
Main Article Content
Abstract
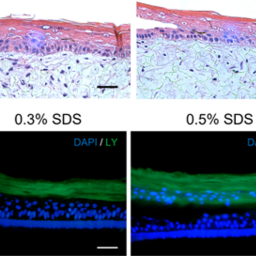
There is a global trend towards the development of physiologically relevant in vitro skin models to reduce or replace animal testing in the evaluation of therapeutic drug candidates. However, only commercial reconstructed human epidermis models (RHEm) have undergone formal validation. Although these commercial models are suitable for a wide range of applications, they are costly, lack flexibility, and the protocols used to generate them are not transparent. In this study, we present an open-source full-thickness skin model (FTSm) and assess its potential for drug testing. The FTSm was developed using endogenous extracellular matrix to recreate the dermal compartment, avoiding animal-derived hydrogels. An RHEm based on an open-source protocol was evaluated in parallel. The integrity of the skin barrier was analyzed by challenging the surface with detergents and measuring cell viability as well as by trans-epithelial electrical resistance (TEER) measurements. Skin irritation studies were performed based on OECD guidelines and complemented with an evaluation of the impact on the skin barrier by TEER measurement. The permeation of a dye through the developed models and a commercial membrane (Strat-M®) was compared using Franz diffusion cells and an infinite dose approach. The FTSm demonstrated structural and barrier properties comparable to native human skin. Although the RHEm showed a better performance in drug testing, the FTSm presented better barrier properties than commercial models as reported in the literature. These skin models can be a valuable contribution to accelerating the development and dissemination of alternatives to animal testing, avoiding the limitations of commercial models.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Ackermann, K., Lombardi Borgia, S., Korting, H. C. et al. (2010). The Phenion® full-thickness skin model for percutaneous absorption testing. Skin Pharmacol Physiol 23, 105-112. doi:10.1159/000265681
Almeida, A., Sarmento, B. and Rodrigues, F. (2017). Insights on in vitro models for safety and toxicity assessment of cosmetic ingredients. Int J Pharm 519, 178-185. doi:10.1016/j.ijpharm.2017.01.024
Carrer, V., Alonso, C., Pont, M. et al. (2020). Effect of propylene glycol on the skin penetration of drugs. Arch Dermatol Res 312, 337-352. doi:10.1007/s00403-019-02017-5
Cartner, T., Brand, N., Tian, K. et al. (2017). Effect of different alcohols on stratum corneum kallikrein 5 and phospholipase A2 together with epidermal keratinocytes and skin irritation. Int J Cosmet Sci 39, 188-196. doi:10.1111/ics.12364
Cotovio, J., Grandidier, M.-H., Portes, P. et al. (2005). The in vitro acute skin irritation of chemicals: Optimisation of the EPISKIN prediction model within the framework of the ECVAM validation process. Altern Lab Anim 33, 329-349. doi:10.1177/026119290503300403
Cross, S. E., Magnusson, B. M., Winckle, G. et al. (2003). Determination of the effect of lipophilicity on the in vitro permeability and tissue reservoir characteristics of topically applied solutes in human skin layers. J Invest Dermatol 120, 759-764. doi:10.1046/j.1523-1747.2003.12131.x
Cui, M., Wiraja, C., Chew, S. W. T. et al. (2021). Nanodelivery systems for topical management of skin disorders. Mol Pharm 18, 491-505. doi:10.1021/acs.molpharmaceut.0c00154
De Vuyst, E., Charlier, C., Giltaire, S. et al. (2013). Reconstruction of normal and pathological human epidermis on polycarbonate filter. In K. Turksen (ed.), Epidermal Cells (191-201). Methods in Molecular Biology (Methods and Protocols), Volume 1195. New York, NY, USA: Springer. doi:10.1007/7651_2013_40
De Wever, B., Kurdykowski, S. and Descargues, P. (2015a). Human skin models for research applications in pharmacology and toxicology: Introducing NativeSkin®, the “missing link” bridging cell culture and/or reconstructed skin models and human clinical testing. Appl In Vitro Toxicol 1, 26-32. doi:10.1089/aivt.2014.0010
De Wever, B., Goldberg, A., Eskes, C. et al. (2015b). “Open source” – Based engineered human tissue models: A new gold standard for nonanimal testing through openness, transparency, and collaboration, promoted by the ALEXANDRA association. Appl In Vitro Toxicol 1, 5-9. doi:10.1089/aivt.2014.0011
Del Bino, S., Duval, C. and Bernerd, F. (2018). Clinical and biological characterization of skin pigmentation diversity and its consequences on UV impact. Int J Mol Sci 19, 2668. doi:10.3390/ijms19092668
Fuggetta, A. (2003). Open source software – An evaluation. J Syst Softw 66, 77-90. doi:10.1016/S0164-1212(02)00065-1
Gorzelanny, C., Mess, C., Schneider, S. W. et al. (2020). Skin barriers in dermal drug delivery: Which barriers have to be overcome and how can we measure them? Pharmaceutics 12, 684. doi:10.3390/pharmaceutics12070684
Gray, A. C., Sidhu, S. S., Chandrasekera, P. C. et al. (2016). Animal-friendly affinity reagents: Replacing the needless in the haystack. Trends Biotechnol 34, 960-969. doi:10.1016/j.tibtech.2016.05.017
Groeber, F., Engelhardt, L., Egger, S. et al. (2015). Impedance spectroscopy for the non-destructive evaluation of in vitro epidermal models. Pharm Res 32, 1845-1854. doi:10.1007/s11095-014-1580-3
Jírová, D., Basketter, D., Liebsch, M. et al. (2010). Comparison of human skin irritation patch test data with in vitro skin irritation assays and animal data. Contact Dermatitis 62, 109-116. doi:10.1111/j.1600-0536.2009.01640.x
Joffe, R., Plaza, J. A. and Kajoian, A. (2020). Tip chapter: Histology and physiology of the skin. In A. Da Costa (ed.), Minimally Invasive Aesthetic Procedures: A Guide for Dermatologists and Plastic Surgeons (179-192). Cham, Switzerland: Springer. doi.org/10.1007/978-3-319-78265-2
Jung, K.-M., Lee, S.-H., Jang, W.-H. et al. (2014). KeraSkin™-VM: A novel reconstructed human epidermis model for skin irritation tests. Toxicol In Vitro 28, 742-750. doi:10.1016/j.tiv.2014.02.014
Mewes, K. R., Fischer, A., Zöller, N. N. et al. (2016). Catch-up validation study of an in vitro skin irritation test method based on an open source reconstructed epidermis (phase I). Toxicol In Vitro 36, 238-253. doi:10.1016/j.tiv.2016.07.007
Natsuga, K., Watanabe, M., Nishie, W. et al. (2019). Life before and beyond blistering: The role of collagen XVII in epidermal physiology. Exp Dermatol 28, 1135-1141. doi:10.1111/exd.13550
Netzlaff, F., Lehr, C. M., Wertz, P. W. et al. (2005). The human epidermis models EpiSkin®, SkinEthic® and EpiDerm®: An evaluation of morphology and their suitability for testing phototoxicity, irritancy, corrosivity, and substance transport. Eur J Pharm Biopharm 60, 167-178. doi:10.1016/j.ejpb.2005.03.004
Niehues, H., Bouwstra, J. A., El Ghalbzouri, A. et al. (2018). 3D skin models for 3R research: The potential of 3D reconstructed skin models to study skin barrier function. Exp Dermatol 27, 501-511. doi:10.1111/exd.13531
OECD (2021). Test No. 439: In Vitro Skin Irritation: Reconstructed Human Epidermis Test Method. OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing, Paris. doi:10.1787/9789264242845-en
Perticaroli, S., Yeomans, D. J., Wireko, F. C. et al. (2019). Translating chemometric analysis into physiological insights from in vivo confocal Raman spectroscopy of the human stratum corneum. Biochim Biophys Acta Biomembr 1861, 403-409. doi:10.1016/j.bbamem.2018.11.007
Radhakrishnan, S., Nagarajan, S., Bechelany, M. et al. (2020). Collagen based biomaterials for tissue engineering applications: A review. In O. Frank-Kamenetskaya, D. Vlasov, E. G. Panova et al. (eds.), Processes and Phenomena on the Boundary Between Biogenic and Abiogenic Nature (3-22). Cham, Switzerland: Springer International Publishing.
Rasmussen, C., Gratz, K., Liebel, F. et al. (2010). The StrataTest® human skin model, a consistent in vitro alternative for toxicological testing. Toxicol In Vitro 24, 2021-2029. doi:10.1016/j.tiv.2010.07.027
Redden, R. A. and Doolin, E. J. (2003). Collagen crosslinking and cell density have distinct effects on fibroblast-mediated contraction of collagen gels. Ski Res Technol 9, 290-293. doi:10.1034/j.1600-0846.2003.00023.x
Rice, G. and Rompolas, P. (2020). Advances in resolving the heterogeneity and dynamics of keratinocyte differentiation. Curr Opin Cell Biol 67, 92-98. doi:10.1016/j.ceb.2020.09.004
Schmook, F. P., Meingassner, J. G. and Billich, A. (2001). Comparison of human skin or epidermis models with human and animal skin in in-vitro percutaneous absorption. Int J Pharm 215, 51-56. doi:10.1016/S0378-5173(00)00665-7
Seo, S. R., Lee, S. G., Lee, H. J. et al. (2017). Disrupted skin barrier is associated with burning sensation after topical tacrolimus application in atopic dermatitis. Acta Derm Venereol 97, 957-958. doi:10.2340/00015555-2699
Seth, D., Cheldize, K., Brown, D. et al. (2017). Global burden of skin disease: Inequities and innovations. Curr Dermatol Rep 6, 204-210. doi:10.1007/s13671-017-0192-7
Srinivasan, B., Kolli, A. R., Esch, M. B. et al. (2015). TEER measurement techniques for in vitro barrier model systems. J Lab Autom 20, 107-126. doi:10.1177/2211068214561025
Tadros, A. R., Romanyuk, A., Miller, I. C. et al. (2020). STAR particles for enhanced topical drug and vaccine delivery. Nat Med 26, 341-347. doi:10.1038/s41591-020-0787-6
Thakoersing, V. S., van Smeden, J., Mulder, A. A. et al. (2013). Increased presence of monounsaturated fatty acids in the stratum corneum of human skin equivalents. J Invest Dermatol 133, 59-67. doi:10.1038/jid.2012.262
Uchida, T., Kadhum, W. R., Kanai, S. et al. (2015). Prediction of skin permeation by chemical compounds using the artificial membrane, Strat-M™. Eur J Pharm Sci 67, 113-118. doi:10.1016/j.ejps.2014.11.002
Urciuolo, F., Casale, C., Imparato, G. et al. (2019). Bioengineered skin substitutes: The role of extracellular matrix and vascularization in the healing of deep wounds. J Clin Med 8, 2083. doi:10.3390/jcm8122083
van den Broek, L. J., Bergers, L. I. J. C., Reijnders, C. M. A. et al. (2017). Progress and future prospectives in skin-on-chip development with emphasis on the use of different cell types and technical challenges. Stem Cell Rev Rep 13, 418-429. doi:10.1007/s12015-017-9737-1
Van Norman, G. A. (2019). Limitations of animal studies for predicting toxicity in clinical trials: Is it time to rethink our current approach? JACC Basic Transl Sci 4, 845-854. doi:10.1016/j.jacbts.2019.10.008
Wang, J. X., Fukunaga-Kalabis, M. and Herlyn, M. (2016). Crosstalk in skin: Melanocytes, keratinocytes, stem cells, and melanoma. J Cell Commun Signal 10, 191-196. doi:10.1007/s12079-016-0349-3
Yamada, M. and Prow, T. W. (2020). Physical drug delivery enhancement for aged skin, UV damaged skin and skin cancer: Translation and commercialization. Adv Drug Deliv Rev 153, 2-17. doi:10.1016/j.addr.2020.04.008
Zoio, P., Ventura, S., Leite, M. et al. (2021a). Pigmented full-thickness human skin model based on a fibroblast-derived matrix for long-term studies. Tissue Eng Part C Methods 27, 433-443. doi:10.1089/ten.tec.2021.0069
Zoio, P., Lopes-Ventura, S. and Oliva, A. (2021b). Barrier-on-a-chip with a modular architecture and integrated sensors for real-time measurement of biological barrier function. Micromachines 12, 816. doi:10.3390/mi12070816
Zoio, P., Lopes-Ventura, S. and Oliva, A. (2022). Biomimetic full-thickness skin-on-a-chip based on a fibroblast-derived matrix. Micro 2, 191-211. doi:10.3390/micro2010013
Zuang, V., Desprez, B., Viegas Barroso, J. et al. (2015). EURL ECVAM Status Report on the Development, Validation and Regulatory Acceptance of Alternative Methods and Approaches. EUR 27474. Luxembourg (Luxembourg): Publications Office of the European Union. JRC97811. doi:10.2788/62058