Perspectives on the evaluation and adoption of complex in vitro models in drug development: Workshop with the FDA and the pharmaceutical industry (IQ MPS Affiliate)
Main Article Content
Abstract
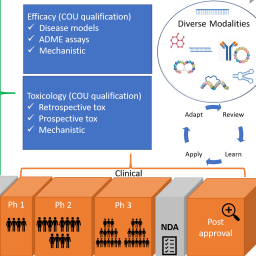
Complex in vitro models (CIVM) offer the potential to improve pharmaceutical clinical drug attrition due to safety and/ or efficacy concerns. For this technology to have an impact, the establishment of robust characterization and qualification plans constructed around specific contexts of use (COU) is required. This article covers the output from a workshop between the Food and Drug Administration (FDA) and Innovation and Quality Microphysiological Systems (IQ MPS) Affiliate. The intent of the workshop was to understand how CIVM technologies are currently being applied by pharmaceutical companies during drug development and are being tested at the FDA through various case studies in order to identify hurdles (real or perceived) to the adoption of microphysiological systems (MPS) technologies, and to address evaluation/qualification pathways for these technologies. Output from the workshop includes the alignment on a working definition of MPS, a detailed description of the eleven CIVM case studies presented at the workshop, in-depth analysis, and key take aways from breakout sessions on ADME (absorption, distribution, metabolism, and excretion), pharmacology, and safety that covered topics such as qualification and performance criteria, species differences and concordance, and how industry can overcome barriers to regulatory submission of CIVM data. In conclusion, IQ MPS Affiliate and FDA scientists were able to build a general consensus on the need for animal CIVMs for preclinical species to better determine species concordance. Furthermore, there was acceptance that CIVM technologies for use in ADME, pharmacology and safety assessment will require qualification, which will vary depending on the specific COU.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Ainslie, G. R., Davis, M., Ewart, L. et al. (2019). Microphysiological lung models to evaluate the safety of new pharmaceutical modalities: A biopharmaceutical perspective. Lab Chip 19, 3152-3161. doi:10.1039/c9lc00492k
Ballard, T. E., Orozco, C. C. and Obach, R. S. (2014). Generation of major human excretory and circulating drug metabolites using a hepatocyte relay method. Drug Metab Dispos 42, 899-902. doi:10.1124/dmd.114.057026
Ballard, T. E., Wang, S., Cox, L. M. et al. (2016). Application of a micropatterned cocultured hepatocyte system to predict preclinical and human-specific drug metabolism. Drug Metab Dispos 44, 172-179. doi:10.1124/dmd.115.066688
Baudy, A. R., Otieno, M. A., Hewitt, P. et al. (2020). Liver microphysiological systems development guidelines for safety risk assessment in the pharmaceutical industry. Lab Chip 20, 215-225. doi:10.1039/c9lc00768g
Bell, C. C., Dankers, A. C. A., Lauschke, V. M. et al. (2018). Comparison of hepatic 2D sandwich cultures and 3D spheroids for long-term toxicity applications: A multicenter study. Toxicol Sci 162, 655-666. doi:10.1093/toxsci/kfx289
Chang, S. Y., Voellinger, J. L., Van Ness, K. P. et al. (2017). Characterization of rat or human hepatocytes cultured in microphysiological systems (MPS) to identify hepatotoxicity. Toxicol In Vitro 40, 170-183. doi:10.1016/j.tiv.2017.01.007
Chen, M., Vijay, V., Shi, Q. et al. (2011). FDA-approved drug labeling for the study of drug-induced liver injury. Drug Discov Today 16, 697-703. doi:10.1016/j.drudis.2011.05.007
Chen, M., Suzuki, A., Thakkar, S. et al. (2016). Dilirank: The largest reference drug list ranked by the risk for developing drug-induced liver injury in humans. Drug Discov Today 21, 648-653. doi:10.1016/j.drudis.2016.02.015
Cohen, S. M. (2004). Human carcinogenic risk evaluation: An alternative approach to the two-year rodent bioassay. Toxicol Sci 80, 225-229. doi:10.1093/toxsci/kfh159
Dalvie, D., Obach, R. S., Kang, P. et al. (2009). Assessment of three human in vitro systems in the generation of major human excretory and circulating metabolites. Chem Res Toxicol 22, 357-368. doi:10.1021/tx8004357
Dame, K. and Ribeiro, A. J. (2021). Microengineered systems with iPSC-derived cardiac and hepatic cells to evaluate drug adverse effects. Exp Biol Med (Maywood) 246, 317-331. doi:10.1177/1535370220959598
Dash, A. and Proctor, W. R. (2019). Chapter 6 – Hepatic microphysiological systems: Current and future applications in drug discovery and development. In J. T. Borenstein, V. Tandon, S. L. Tao et al. (eds), Microfluidic Cell Culture Systems (159-186). 2nd edition. Elsevier. doi:10.1016/B978-0-12-813671-3.00006-2
Deosarkar, S. P., Prabhakarpandian, B., Wang, B. et al. (2015). A novel dynamic neonatal blood-brain barrier on a chip. PLoS One 10, e0142725. doi:10.1371/journal.pone.0142725
Dolmetsch, R. and Geschwind, D. H. (2011). The human brain in a dish: The promise of iPSC-derived neurons. Cell 145, 831-834. doi:10.1016/j.cell.2011.05.034
Easter, A., Bell, M. E., Damewood, J. R., Jr. et al. (2009). Approaches to seizure risk assessment in preclinical drug discovery. Drug Discov Today 14, 876-884. doi:10.1016/j.drudis.2009.06.003
Ekert, J. E., Deakyne, J., Pribul-Allen, P. et al. (2020). Recommended guidelines for developing, qualifying, and implementing complex in vitro models (CIVMs) for drug discovery. SLAS Discov 25, 1174-1190. doi:10.1177/2472555220923332
Ewart, L., Fabre, K., Chakilam, A. et al. (2017). Navigating tissue chips from development to dissemination: A pharmaceutical industry perspective. Exp Biol Med (Maywood) 242, 1579-1585. doi:10.1177/1535370217715441
Fabre, K., Berridge, B., Proctor, W. R. et al. (2020). Introduction to a manuscript series on the characterization and use of microphysiological systems (MPS) in pharmaceutical safety and ADME applications. Lab Chip 20, 1049-1057. doi:10.1039/c9lc01168d
FDA (2021a). https://www.fda.gov/drugs/development-approval-process-drugs/drug-development-tool-ddt-qualification-programs
FDA (2021b). https://www.fda.gov/vaccines-blood-biologics/science-research-biologics/innovation-and-regulatory-science
Fermini, B., Coyne, S. T. and Coyne, K. P. (2018). Clinical trials in a dish: A perspective on the coming revolution in drug development. SLAS Discov 23, 765-776. doi:10.1177/2472555218775028
Fowler, S., Chen, W. L. K., Duignan, D. B. et al. (2020). Microphysiological systems for ADME-related applications: Current status and recommendations for system development and characterization. Lab Chip 20, 446-467. doi:10.1039/c9lc00857h
Grunwald, L. M., Stock, R., Haag, K. et al. (2019). Comparative characterization of human induced pluripotent stem cells (hiPSC) derived from patients with schizophrenia and autism. Transl Psychiatry 9, 179. doi:10.1038/s41398-019-0517-3
Hardwick, R. N., Betts, C. J., Whritenour, J. et al. (2020). Drug-induced skin toxicity: Gaps in preclinical testing cascade as opportunities for complex in vitro models and assays. Lab Chip 20, 199-214. doi:10.1039/c9lc00519f
Hendrix, S. B., Mogg, R., Wang, S. J. et al. (2021). Perspectives on statistical strategies for the regulatory biomarker qualification process. Biomark Med 15, 669-684. doi:10.2217/bmm-2020-0523
Huh, D., Leslie, D. C., Matthews, B. D. et al. (2012). A human disease model of drug toxicity-induced pulmonary edema in a lung-on-a-chip microdevice. Sci Transl Med 4, 159ra147. doi:10.1126/scitranslmed.3004249
Isoherranen, N., Madabushi, R. and Huang, S. M. (2019). Emerging role of organ-on-a-chip technologies in quantitative clinical pharmacology evaluation. Clin Transl Sci 12, 113-121. doi:10.1111/cts.12627
Kang, W., Podtelezhnikov, A. A., Tanis, K. Q. et al. (2020). Development and application of a transcriptomic signature of bioactivation in an advanced in vitro liver model to reduce drug-induced liver injury risk early in the pharmaceutical pipeline. Toxicol Sci 177, 121-139. doi:10.1093/toxsci/kfaa094
Khetani, S. R. and Bhatia, S. N. (2008). Microscale culture of human liver cells for drug development. Nat Biotechnol 26, 120-126. doi:10.1038/nbt1361
Khetani, S. R., Berger, D. R., Ballinger, K. R. et al. (2015). Microengineered liver tissues for drug testing. J Lab Autom 20, 216-250. doi:10.1177/2211068214566939
Lam, J., Bellayr, I. H., Marklein, R. A. et al. (2018). Functional profiling of chondrogenically induced multipotent stromal cell aggregates reveals transcriptomic and emergent morphological phenotypes predictive of differentiation capacity. Stem Cells Transl Med 7, 664-675. doi:10.1002/sctm.18-0065
Lancaster, M. A. and Knoblich, J. A. (2014). Organogenesis in a dish: Modeling development and disease using organoid technologies. Science 345, 1247125. doi:10.1126/science.1247125
Leptak, C., Menetski, J. P., Wagner, J. A. et al. (2017). What evidence do we need for biomarker qualification? Sci Transl Med 9, eaal4599. doi:10.1126/scitranslmed.aal4599
Low, L. A. and Tagle, D. A. (2017). Tissue chips – Innovative tools for drug development and disease modeling. Lab Chip 17, 3026-3036. doi:10.1039/c7lc00462a
Malki, M. A. and Pearson, E. R. (2020). Drug-drug-gene interactions and adverse drug reactions. Pharmacogenomics J 20, 355-366. doi:10.1038/s41397-019-0122-0
Mannhardt, I., Saleem, U., Mosqueira, D. et al. (2020). Comparison of 10 control hPSC lines for drug screening in an engineered heart tissue format. Stem Cell Reports 15, 983-998. doi:10.1016/j.stemcr.2020.09.002
Mao, A. S. and Mooney, D. J. (2015). Regenerative medicine: Current therapies and future directions. Proc Natl Acad Sci U S A 112, 14452-14459. doi:10.1073/pnas.1508520112
Marklein, R. A., Lam, J., Guvendiren, M. et al. (2018). Functionally-relevant morphological profiling: A tool to assess cellular heterogeneity. Trends Biotechnol 36, 105-118. doi:10.1016/j.tibtech.2017.10.007
Mead, A. N., Amouzadeh, H. R., Chapman, K. et al. (2016). Assessing the predictive value of the rodent neurofunctional assessment for commonly reported adverse events in phase I clinical trials. Regul Toxicol Pharmacol 80, 348-357. doi:10.1016/j.yrtph.2016.05.002
Nguyen, D. G. and Pentoney, S. L., Jr. (2017). Bioprinted three dimensional human tissues for toxicology and disease modeling. Drug Discov Today Technol 23, 37-44. doi:10.1016/j.ddtec.2017.03.001
Ohbuchi, M., Tetsuka, K., Nagata, M. et al. (2018). Mechanistic study of acetaminophen-induced liver injury using a 3D bioprinted human liver tissue model. Human and Animal Bridging Research Organization Academic Annual Meeting Programm / Abstracts 25, 90.
Perez-Terzic, C. and Childers, M. K. (2014). Regenerative rehabilitation: A new future? Am J Phys Med Rehabil 93, S73-78. doi:10.1097/PHM.0000000000000211
Peters, M. F., Choy, A. L., Pin, C. et al. (2020). Developing in vitro assays to transform gastrointestinal safety assessment: Potential for microphysiological systems. Lab Chip 20, 1177-1190. doi:10.1039/c9lc01107b
Peterson, N. C., Mahalingaiah, P. K., Fullerton, A. et al. (2020). Application of microphysiological systems in biopharmaceutical research and development. Lab Chip 20, 697-708. doi:10.1039/c9lc00962k
Phillips, J. A., Grandhi, T. S. P., Davis, M. et al. (2020). A pharmaceutical industry perspective on microphysiological kidney systems for evaluation of safety for new therapies. Lab Chip 20, 468-476. doi:10.1039/c9lc00925f
Pointon, A., Maher, J., Davis, M. et al. (2021). Cardiovascular microphysiological systems (CVMPS) for safety studies – A pharma perspective. Lab Chip 21, 458-472. doi:10.1039/d0lc01040e
Proctor, W. R., Foster, A. J., Vogt, J. et al. (2017). Utility of spherical human liver microtissues for prediction of clinical drug-induced liver injury. Arch Toxicol 91, 2849-2863. doi:10.1007/s00204-017-2002-1
Ribeiro, A. J. S., Guth, B. D., Engwall, M. et al. (2019a). Considerations for an in vitro, cell-based testing platform for detection of drug-induced inotropic effects in early drug development. Part 2: Designing and fabricating microsystems for assaying cardiac contractility with physiological relevance using human iPSC-cardiomyocytes. Front Pharmacol 10, 934. doi:10.3389/fphar.2019.00934
Ribeiro, A. J. S., Yang, X., Patel, V. et al. (2019b). Liver microphysiological systems for predicting and evaluating drug effects. Clin Pharmacol Ther 106, 139-147. doi:10.1002/cpt.1458
Rouse, R., Kruhlak, N., Weaver, J. et al. (2018a). Translating new science into the drug review process: The US FDA’s division of applied regulatory science. Ther Innov Regul Sci 52, 244-255. doi:10.1177/2168479017720249
Rouse, R., Zineh, I. and Strauss, D. G. (2018b). Regulatory science – An underappreciated component of translational research. Trends Pharmacol Sci 39, 225-229. doi:10.1016/j.tips.2017.12.006
Rubiano, A., Indapurkar, A., Yokosawa, R. et al. (2021). Characterizing the reproducibility in using a liver microphysiological system for assaying drug toxicity, metabolism, and accumulation. Clin Transl Sci 14, 1049-1061. doi:10.1111/cts.12969
Sirenko, O., Parham, F., Dea, S. et al. (2019). Functional and mechanistic neurotoxicity profiling using human iPSC-derived neural 3D cultures. Toxicol Sci 167, 58-76. doi:10.1093/toxsci/kfy218
Stresser, D. M., Sun, J. and Wilson, S. S. (2021). Evaluation of tissue stem cell-derived human intestinal organoids, a physiologically relevant model to evaluate cytochrome P450 induction in gut. Drug Metab Dispos 49, 245-253. doi:10.1124/dmd.120.000281
Sung, K. E., Arcidiacono, J., Fink, D. W. et al. (2020). Chapter 84 – The regulatory process from concept to market. In R. Lanza, R. Langer, J. P. Vacanti et al. (eds.), Principles of Tissue Engineering (1553-1572). 5th edition. Academic Press. doi:10.1016/B978-0-12-818422-6.00086-1
Tetsuka, K., Ohbuchi, M., Kawabe, T. et al. (2020). Reconstituted human organ models as a translational tool for human organ response: Definition, expectations, cases, and strategies for implementation in drug discovery and development. Biol Pharm Bull 43, 375-383. doi:10.1248/bpb.b19-01070
Van Vleet, T. R., Liguori, M. J., Lynch, J. J., 3rd et al. (2019). Screening strategies and methods for better off-target liability prediction and identification of small-molecule pharmaceuticals. SLAS Discov 24, 1-24. doi:10.1177/2472555218799713
Weaver, R. J. and Valentin, J. P. (2019). Today’s challenges to de-risk and predict drug safety in human “mind-the-gap”. Toxicol Sci 167, 307-321. doi:10.1093/toxsci/kfy270
Wilson, W. C., Jr. and Boland, T. (2003). Cell and organ printing 1: Protein and cell printers. Anat Rec A Discov Mol Cell Evol Biol 272, 491-496. doi:10.1002/ar.a.10057