A strategy towards the generation of testable adverse outcome pathways for nanomaterials
Main Article Content
Abstract
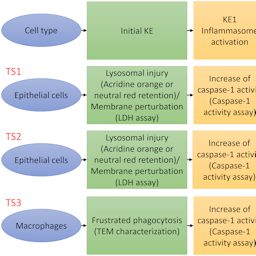
Manufactured nanomaterials (NMs) are increasingly used in a wide range of industrial applications leading to a constant increase in the market size of nano-enabled products. The increased production and use of NMs are raising concerns among different stakeholder groups with regard to their effects on human and environmental health. Currently, nanosafety hazard assessment is still widely performed using in vivo (animal) models, however the development of robust and regulatory relevant strategies is required to prioritize and/or reduce animal testing. An adverse outcome pathway (AOP) is a structured representation of biological events that start from a molecular initiating event (MIE) leading to an adverse outcome (AO) through a series of key events (KEs). The AOP framework offers great advancement to risk assessment and regulatory safety assessments. While AOPs for chemicals have been more frequently reported, the AOP collection for NMs is limited. By using existing AOPs, we aimed to generate simple and testable strategies to predict if a given NM has the potential to induce a MIE leading to an AO through a series of KEs. Firstly, we identified potential MIEs or initial KEs reported for NMs in the literature. Then, we searched the identified MIE or initial KEs as keywords in the AOP-Wiki to find associated AOPs. Finally, using two case studies, we demonstrate how in vitro strategies can be used to test the identified MIE/KEs.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Allen, T. E. H., Goodman, J. M., Gutsell, S. et al. (2014). Defining molecular initiating events in the adverse outcome pathway framework for risk assessment. Chem Res Toxicol 27, 2100–2112. https://doi.org/10.1021/tx500345j.
Ankley, G. T., Bennett, R. S., Erickson, R. J. et al. (2010). Adverse outcome pathways: A conceptual framework to support ecotoxicology research and risk assessment. Environ Toxicol Chem 29, 730–741. https://doi.org/10.1002/etc.34.
Annangi, B., Rubio, L., Alaraby, M. et al. (2016). Acute and long-term in vitro effects of zinc oxide nanoparticles. Arch Toxicol 90, 2201–2213. https://doi.org/10.1007/s00204-015-1613-7.
AOP-Wiki Activation, Stellate cells leads to Accumulation, Collagen, KER 295. Available at: https://aopwiki.org/relationships/295 [Accessed February 14, 2021a].
AOP-Wiki Tissue resident cell activation, KE 1492. Available at: https://aopwiki.org/events/1492 [Accessed February 14, 2021b].
Braakhuis, H. M., Gosens, I., Heringa, M. B. et al. (2021). Mechanism of Action of TiO 2 : Recommendations to Reduce Uncertainties Related to Carcinogenic Potential . Annu Rev Pharmacol Toxicol 61, 1–21. https://doi.org/10.1146/annurev-pharmtox-101419-100049.
Brand, W., Peters, R. J. B., Braakhuis, H. M. et al. (2020). Possible effects of titanium dioxide particles on human liver, intestinal tissue, spleen and kidney after oral exposure. Nanotoxicology 14, 985–1007. https://doi.org/10.1080/17435390.2020.1778809.
Chen, N., Song, Z. M., Tang, H. et al. (2016). Toxicological effects of Caco-2 cells following short-term and long-term exposure to Ag nanoparticles. Int J Mol Sci 17. https://doi.org/10.3390/ijms17060974.
Choi, J. Y., Ramachandran, G. and Kandlikar, M. (2009). The impact of toxicity testing costs on nanomaterial regulation. Environ Sci Technol 43, 3030–3034. https://doi.org/10.1021/es802388s.
Collins, A. R., Annangi, B., Rubio, L. et al. (2017). High throughput toxicity screening and intracellular detection of nanomaterials. Wiley Interdiscip Rev Nanomedicine Nanobiotechnology 9. https://doi.org/10.1002/wnan.1413.
Delrue, N., Sachana, M., Sakuratani, Y. et al. (2016). The adverse outcome pathway concept: A basis for developing regulatory Decision-making tools. ATLA Altern to Lab Anim 44, 417–429. https://doi.org/10.1177/026119291604400504.
Drasler, B., Sayre, P., Steinhäuser, K. G. et al. (2017). In vitro approaches to assess the hazard of nanomaterials. NanoImpact 8, 99–116. https://doi.org/10.1016/j.impact.2017.08.002.
Ede, J. D., Lobaskin, V., Vogel, U. et al. (2020). Translating Scientific Advances in the AOP Framework to Decision Making for Nanomaterials. Nanomaterials 10. https://doi.org/10.3390/nano10061229.
European Commission (2020). Animals used for scientific purposes. Available at: https://ec.europa.eu/environment/chemicals/lab_animals/index_en.htm [Accessed December 18, 2020].
Gajewicz, A., Schaeublin, N., Rasulev, B. et al. (2015). Towards understanding mechanisms governing cytotoxicity of metal oxides nanoparticles: Hints from nano-QSAR studies. Nanotoxicology 9, 313–325. https://doi.org/10.3109/17435390.2014.930195.
Gerloff, K., Landesmann, B., Worth, A. et al. (2017). The Adverse Outcome Pathway approach in nanotoxicology. Comput Toxicol 1, 3–11. https://doi.org/10.1016/j.comtox.2016.07.001.
Guadagnini, R., Halamoda Kenzaoui, B., Walker, L. et al. (2015). Toxicity screenings of nanomaterials: Challenges due to interference with assay processes and components of classic in vitro tests. Nanotoxicology 9, 13–24. https://doi.org/10.3109/17435390.2013.829590.
Halappanavar, S., Van Den Brule, S., Nymark, P. et al. (2020). Adverse outcome pathways as a tool for the design of testing strategies to support the safety assessment of emerging advanced materials at the nanoscale. Part Fibre Toxicol 17, 1–24. https://doi.org/10.1186/s12989-020-00344-4.
Halappanavar, S., Ede, J. D., Shatkin, J. A. et al. (2019a). A systematic process for identifying key events for advancing the development of nanomaterial relevant adverse outcome pathways. NanoImpact 15, 100178. https://doi.org/10.1016/j.impact.2019.100178.
Halappanavar, S., Ede, J. D., Shatkin, J. A. et al. (2019b). A systematic process for identifying key events for advancing the development of nanomaterial relevant adverse outcome pathways. NanoImpact 15. https://doi.org/10.1016/j.impact.2019.100178.
Halappanavar, S., Nymark, P., Krug, H. F. et al. (2021). Non-Animal Strategies for Toxicity Assessment of Nanoscale Materials : Role of Adverse Outcome Pathways in the Selection of Endpoints. 2007628. https://doi.org/10.1002/smll.202007628.
Hoet, P. H., Nemery, B. and Napierska, D. (2013). Intracellular oxidative stress caused by nanoparticles: What do we measure with the dichlorofluorescein assay? Nano Today 8, 223–227. https://doi.org/10.1016/j.nantod.2013.01.001.
ISO - ISO/IEC Guide 98-3 (2008). ISO - ISO/IEC Guide 98-3:2008 - Uncertainty of measurement — Part 3: Guide to the expression of uncertainty in measurement (GUM:1995). Available at: https://www.iso.org/standard/50461.html [Accessed February 19, 2021].
Jagiello, K., Halappanavar, S., Rybińska‐Fryca, A. et al. (2021). Transcriptomics‐Based and AOP‐Informed Structure–Activity Relationships to Predict Pulmonary Pathology Induced by Multiwalled Carbon Nanotubes. Small, 2003465. https://doi.org/10.1002/smll.202003465.
Kroll, A., Dierker, C., Rommel, C. et al. (2011). Cytotoxicity screening of 23 engineered nanomaterials using a test matrix of ten cell lines and three different assays. Part Fibre Toxicol 8. https://doi.org/10.1186/1743-8977-8-9.
Kroll, A., Pillukat, M. H., Hahn, D. et al. (2012). Interference of engineered nanoparticles with in vitro toxicity assays. Arch Toxicol 86, 1123–1136. https://doi.org/10.1007/s00204-012-0837-z.
Kubinyi, H. (1997). QSAR and 3D QSAR in drug design. Part 1: Methodology. Drug Discov Today 2, 457–467. https://doi.org/10.1016/S1359-6446(97)01079-9.
Li, J. T., Liao, Z. X., Ping, J. et al. (2008). Molecular mechanism of hepatic stellate cell activation and antifibrotic therapeutic strategies. J Gastroenterol 43, 419–428. https://doi.org/10.1007/s00535-008-2180-y.
Liu, T., Wang, X., Karsdal, M. A. et al. (2012). Molecular serum markers of liver fibrosis. Biomark Insights 7, 105–117. https://doi.org/10.4137/BMI.S10009.
Murugadoss, S., Lison, D., Godderis, L. et al. (2017). Toxicology of silica nanoparticles: an update. Arch Toxicol 91, 2967–3010. https://doi.org/10.1007/s00204-017-1993-y.
OECD (2015). PERFORMANCE STANDARDS FOR THE ASSESSMENT OF PROPOSED SIMILAR OR MODIFIED IN VITRO RECONSTRUCTED HUMAN EPIDERMIS (RhE) TEST METHODS FOR SKIN IRRITATION TESTING AS DESCRIBED IN TG 439. Available at: http://www.oecd.org/officialdocuments/publicdisplaydocumentpdf/?cote=ENV/JM/MONO(2015)27&doclanguage=en [Accessed February 19, 2021].
OECD (2020). REPORT ON CONSIDERATIONS FROM CASE STUDIES ON INTEGRATED APPROACHES. Available at: http://www.oecd.org/officialdocuments/publicdisplaydocumentpdf/?cote=env/jm/mono(2020)24&doclanguage=en [Accessed February 19, 2021].
Ostermann, M., Sauter, A., Xue, Y. et al. (2020). Label-free impedance flow cytometry for nanotoxicity screening. Sci Rep 10, 1–14. https://doi.org/10.1038/s41598-019-56705-3.
Pan, Y., Li, T., Cheng, J. et al. (2016). Nano-QSAR modeling for predicting the cytotoxicity of metal oxide nanoparticles using novel descriptors. RSC Adv 6, 25766–25775. https://doi.org/10.1039/c6ra01298a.
Puzyn, T., Rasulev, B., Gajewicz, A. et al. (2011). Using nano-QSAR to predict the cytotoxicity of metal oxide nanoparticles. Nat Nanotechnol 6, 175–178. https://doi.org/10.1038/nnano.2011.10.
Ruzycka, M., Cimpan, M. R., Rios-Mondragon, I. et al. (2019). Microfluidics for studying metastatic patterns of lung cancer. J Nanobiotechnology 17, 1–30. https://doi.org/10.1186/s12951-019-0492-0.
Sachana, M., Rolaki, A. and Bal-Price, A. (2018). Development of the Adverse Outcome Pathway (AOP): Chronic binding of antagonist to N-methyl-D-aspartate receptors (NMDARs) during brain development induces impairment of learning and memory abilities of children. Toxicol Appl Pharmacol 354, 153–175. https://doi.org/10.1016/j.taap.2018.02.024.
Savolainen, K., Alenius, H., Norppa, H. et al. (2010). Risk assessment of engineered nanomaterials and nanotechnologies-A review. Toxicology 269, 92–104. https://doi.org/10.1016/j.tox.2010.01.013.
Seiffert, J. M., Baradez, M. O., Nischwitz, V. et al. (2012). Dynamic monitoring of metal oxide nanoparticle toxicity by label free impedance sensing. Chem Res Toxicol 25, 140–152. https://doi.org/10.1021/tx200355m.
Shah, P., Kaushik, A., Zhu, X. et al. (2014). Chip based single cell analysis for nanotoxicity assessment. https://doi.org/10.1039/c3an02280c.
Shi, H., Magaye, R., Castranova, V. et al. (2013). Titanium dioxide nanoparticles: A review of current toxicological data. Part Fibre Toxicol 10. https://doi.org/10.1186/1743-8977-10-15.
Stark, W. J., Stoessel, P. R., Wohlleben, W. et al. (2015). Industrial applications of nanoparticles. Chem Soc Rev 44, 5793–5805. https://doi.org/10.1039/c4cs00362d.
Stern, S. T., Adiseshaiah, P. P. and Crist, R. M. (2012). Autophagy and lysosomal dysfunction as emerging mechanisms of nanomaterial toxicity. Part Fibre Toxicol 9, 20. https://doi.org/10.1186/1743-8977-9-20.
Vance, M. E., Kuiken, T., Vejerano, E. P. et al. (2015). Nanotechnology in the real world: Redeveloping the nanomaterial consumer products inventory. Beilstein J Nanotechnol 6, 1769–1780. https://doi.org/10.3762/bjnano.6.181.
Vietti, G., Lison, D. and van den Brule, S. (2016). Mechanisms of lung fibrosis induced by carbon nanotubes: towards an Adverse Outcome Pathway (AOP). Part Fibre Toxicol 13, 11. https://doi.org/10.1186/s12989-016-0123-y.
Villeneuve, D. L., Crump, D., Garcia-Reyero, N. et al. (2014). Adverse outcome pathway development II: Best practices. Toxicol Sci 142, 321–330. https://doi.org/10.1093/toxsci/kfu200.
Vinković Vrček, I., Pavičić, I., Crnković, T. et al. (2015). Does surface coating of metallic nanoparticles modulate their interference with in vitro assays? RSC Adv 5, 70787–70807. https://doi.org/10.1039/c5ra14100a.
Xi, W. S., Song, Z. M., Chen, Z. et al. (2019). Short-term and long-term toxicological effects of vanadium dioxide nanoparticles on A549 cells. Environ Sci Nano 6, 565–579. https://doi.org/10.1039/C8EN00959G.