Novel prediction models for genotoxicity based on biomarker genes in human HepaRG™ cells
Main Article Content
Abstract
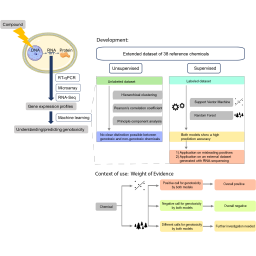
Transcriptomics-based biomarkers are promising new approach methodologies (NAMs) to identify molecular events underlying the genotoxic mode of action of chemicals. Previously, we developed the GENOMARK biomarker, consisting of 84 genes selected based on whole genomics DNA microarray profiles of 24 (non-)genotoxic reference chemicals covering different modes of action in metabolically competent human HepaRG™ cells. In the present study, new prediction models for genotoxicity were developed based on an extended reference dataset of 38 chemicals including existing as well as newly generated gene expression data. Both unsupervised and supervised machine learning algorithms were used, but as unsupervised machine learning did not clearly distinguish between groups, the performance of two supervised machine learning algorithms, i.e., support vector machine (SVM) and random forest (RF), was evaluated. More specifically, the predictive accuracy was compared, the sensitivity to outliers for one or more biomarker genes was assessed, and the prediction performance for 10 misleading positive chemicals exposed at their IC10 concentration was determined. In addition, the applicability of both prediction models on a publicly available gene expression dataset, generated with RNA-sequencing, was investigated. Overall, the RF and SVM models were complementary in their classification of chemicals for genotoxicity. To facilitate data analysis, an online application was developed, combining the outcomes of both prediction models. This research demonstrates that the combination of gene expression data with supervised machine learning algorithms can contribute to the ongoing paradigm shift towards a more human-relevant in vitro genotoxicity testing strategy without the use of experimental animals.
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.
Articles are distributed under the terms of the Creative Commons Attribution 4.0 International license (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium, provided the original work is appropriately cited (CC-BY). Copyright on any article in ALTEX is retained by the author(s).
Ach, R. A., Wang, H. and Curry, B. (2008). Measuring microRNAs: Comparisons of microarray and quantitative PCR measurements, and of different total RNA prep methods. BMC Biotechnol 8, 69. doi:10.1186/1472-6750-8-69
Akoglu, H. (2018). User’s guide to correlation coefficients. Turk J Emerg Med 18, 91-93. doi:10.1016/j.tjem.2018.08.001
Alexander-Dann, B., Pruteanu, L. L., Oerton, E. et al. (2018). Developments in toxicogenomics: Understanding and predicting compound-induced toxicity from gene expression data. Mol Omics 14, 218-236. doi:10.1039/c8mo00042e
Ates, G., Doktorova, T. Y., Pauwels, M. et al. (2014). Retrospective analysis of the mutagenicity/genotoxicity data of the cosmetic ingredients present on the Annexes of the Cosmetic EU legislation (2000-12). Mutagenesis 29, 115-121. doi:10.1093/mutage/get068
Ates, G., Favyts, D., Hendriks, G. et al. (2016a). The Vitotox and ToxTracker assays: A two-test combination for quick and reliable assessment of genotoxic hazards. Mutat Res 810, 13-21. doi:10.1016/j.mrgentox.2016.09.005
Ates, G., Raitano, G., Heymans, A. et al. (2016b). In silico tools and transcriptomics analyses in the mutagenicity assessment of cosmetic ingredients: A proof-of-principle on how to add weight to the evidence. Mutagenesis 31, 453-461. doi:10.1093/mutage/gew008
Ates, G., Mertens, B., Heymans, A. et al. (2018). A novel genotoxin-specific qPCR array based on the metabolically competent human HepaRG™ cell line as a rapid and reliable tool for improved in vitro hazard assessment. Arch Toxicol 92, 1593-1608. doi:10.1007/s00204-018-2172-5
Benesty, J., Chen, J. and Huang, Y. (2008). On the importance of the Pearson correlation coefficient in noise reduction. IEEE Transactions on Audio, Speech, and Language Processing 16, 757-765. doi:10.1109/TASL.2008.919072
Braeuning, A., Vetter, S., Orsetti, S. et al. (2012). Paradoxical cytotoxicity of tert-butylhydroquinone in vitro: What kills the untreated cells? Arch Toxicol 86, 1481-1487. doi:10.1007/s00204-012-0841-3
Breiman, L. (2001). Random forests. Machine Learning 45, 5-32. doi:10.1023/a:1010933404324
Buick, J. K., Williams, A., Gagné, R. et al. (2020). Flow cytometric micronucleus assay and TGx-DDI transcriptomic biomarker analysis of ten genotoxic and non-genotoxic chemicals in human HepaRG™ cells. Genes Environ 42, 5. doi:10.1186/s41021-019-0139-2
Buick, J. K., Williams, A., Meier, M. J. et al. (2021). A modern genotoxicity testing paradigm: Integration of the high-throughput CometChip® and the TGx-DDI transcriptomic biomarker in human HepaRG™ cell cultures. Front Public Health 9, 694834. doi:10.3389/fpubh.2021.694834
Cho, T. M., Rose, R. L. and Hodgson, E. (2006). In vitro metabolism of naphthalene by human liver microsomal cytochrome p450 enzymes. Drug Metab Dispos 34, 176-183. doi:10.1124/dmd.105.005785
Corvi, R. and Madia, F. (2017). In vitro genotoxicity testing – Can the performance be enhanced? Food Chem Toxicol 106, 600-608. doi:10.1016/j.fct.2016.08.024
Dallas, P. B., Gottardo, N. G., Firth, M. J. et al. (2005). Gene expression levels assessed by oligonucleotide microarray analysis and quantitative real-time RT-PCR – How well do they correlate? BMC Genomics 6, 59. doi:10.1186/1471-2164-6-59
David, R. (2020). The promise of toxicogenomics for genetic toxicology: Past, present and future. Mutagenesis 35, 153-159. doi:10.1093/mutage/geaa007
Deist, T. M., Dankers, F. J. W. M., Valdes, G. et al. (2018). Machine learning algorithms for outcome prediction in (chemo)radiotherapy: An empirical comparison of classifiers. Med Phys 45, 3449-3459. doi:10.1002/mp.12967
Dobo, K. L. and Eastmond, D. A. (1994). Role of oxygen radicals in the chromosomal loss and breakage induced by the quinone-forming compounds, hydroquinone and tert-butylhydroquinone. Environ Mol Mutagen 24, 293-300. doi:10.1002/em.2850240406
Doherty, M. D., Cohen, G. M. and Smith, M. T. (1984). Mechanisms of toxic injury to isolated hepatocytes by 1-naphthol. Biochem Pharmacol 33, 543-549. doi:10.1016/0006-2952(84)90305-8
EC (2009). Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on cosmetic products. Off J Eur Union L342, 1-393. http://data.europa.eu/eli/reg/2009/1223/oj
EC – European Commission, Directorate-General for Health and Food Safety (2022). The SCCS notes of guidance for the testing of cosmetic ingredients and their safety evaluation: 11th revision. Publications Office of the European Union. SCCS/1628/21. doi:10.2875/273162
ECHA (2007). Registration Dossier 2-tert-butylhydroquinone. European Chemicals Agency. https://echa.europa.eu/da/registration-dossier/-/registered-dossier/5612/7/7/1
EFSA (2004). Opinion of the scientific panel on food additives, flavourings, processing aids and materials in contact with food (AFC) on a request from the Commission related to tertiary-butylhydroquinone (TBHQ). EFSA J 2, 1-50. doi:10.2903/j.efsa.2004.84
Elder, R. L. (1991). Final report on the safety assessment of hydroxybenzomorpholine. J Am Coll Toxicol 10, 205-213. doi:10.3109/10915819109078630
Fernández-Delgado, M., Cernadas, E., Barro, S. et al. (2014). Do we need hundreds of classifiers to solve real world classification problems? J Mach Learn Res 15, 3133-3181.
Fowler, P., Smith, K., Young, J. et al. (2012). Reduction of misleading (“false”) positive results in mammalian cell genotoxicity assays. I. Choice of cell type. Mutat Res 742, 11-25. doi:10.1016/j.mrgentox.2011.10.014
Fowler, P., Meurer, K., Honarvar, N. et al. (2018). A review of the genotoxic potential of 1,4-naphthoquinone. Mutat Res Genet Toxicol Environ Mutagen 834, 6-17. doi:10.1016/j.mrgentox.2018.07.004
Gerets, H. H., Tilmant, K., Gerin, B. et al. (2012). Characterization of primary human hepatocytes, HepG2 cells, and HepaRG cells at the mRNA level and CYP activity in response to inducers and their predictivity for the detection of human hepatotoxins. Cell Biol Toxicol 28, 69-87. doi:10.1007/s10565-011-9208-4
Gharavi, N., Haggarty, S. and El-Kadi, A. O. (2007). Chemoprotective and carcinogenic effects of tert-butylhydroquinone and its metabolites. Curr Drug Metab 8, 1-7. doi:10.2174/138920007779315035
Kapuci, M., Ulker, Z., Gurkan, S. et al. (2014). Determination of cytotoxic and genotoxic effects of naphthalene, 1-naphthol and 2-naphthol on human lymphocyte culture. Toxicol Ind Health 30, 82-89. doi:10.1177/0748233712451772
Kassambara, A. (2017). Practical Guide to Cluster Analysis in R: Unsupervised Machine Learning. STHDA.
Kirkland, D., Aardema, M., Henderson, L. et al. (2005). Evaluation of the ability of a battery of three in vitro genotoxicity tests to discriminate rodent carcinogens and non-carcinogens I. Sensitivity, specificity and relative predictivity. Mutat Res 584, 1-256. doi:10.1016/j.mrgentox.2005.02.004
Kirkland, D., Pfuhler, S., Tweats, D. et al. (2007). How to reduce false positive results when undertaking in vitro genotoxicity testing and thus avoid unnecessary follow-up animal tests: Report of an ECVAM workshop. Mutat Res 628, 31-55. doi:10.1016/j.mrgentox.2006.11.008
Kirkland, D., Kasper, P., Müller, L. et al. (2008). Recommended lists of genotoxic and non-genotoxic chemicals for assessment of the performance of new or improved genotoxicity tests: A follow-up to an ECVAM workshop. Mutat Res 653, 99-108. doi:10.1016/j.mrgentox.2008.03.008
Kirkland, D., Kasper, P., Martus, H. J. et al. (2016). Updated recommended lists of genotoxic and non-genotoxic chemicals for assessment of the performance of new or improved genotoxicity tests. Mutat Res Genet Toxicol Environ Mutagen 795, 7-30. doi:10.1016/j.mrgentox.2015.10.006
Kuhn, M. (2008). Building predictive models in R using the caret package. J Stat Softw 28, 1-26. doi:10.18637/jss.v028.i05
Lambert, C. B., Spire, C., Claude, N. et al. (2009). Dose- and time-dependent effects of phenobarbital on gene expression profiling in human hepatoma HepaRG cells. Toxicol Appl Pharmacol 234, 345-360. doi:10.1016/j.taap.2008.11.008
Li, H. H., Hyduke, D. R., Chen, R. et al. (2015). Development of a toxicogenomics signature for genotoxicity using a dose-optimization and informatics strategy in human cells. Environ Mol Mutagen 56, 505-519. doi:10.1002/em.21941
Livak, K. J. and Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔC(T) method. Methods 25, 402-408. doi:10.1006/meth.2001.1262
Magkoufopoulou, C., Claessen, S. M., Tsamou, M. et al. (2012). A transcriptomics-based in vitro assay for predicting chemical genotoxicity in vivo. Carcinogenesis 33, 1421-1429. doi:10.1093/carcin/bgs182
Merrick, B. A. (2019). Next generation sequencing data for use in risk assessment. Curr Opin Toxicol 18, 18-26. doi:10.1016/j.cotox.2019.02.010
Miller, M. G., Rodgers, A. and Cohen, G. M. (1986). Mechanisms of toxicity of naphthoquinones to isolated hepatocytes. Biochem Pharmacol 35, 1177-1184. doi:10.1016/0006-2952(86)90157-7
Mišík, M., Nersesyan, A., Ropek, N. et al. (2019). Use of human derived liver cells for the detection of genotoxins in comet assays. Mutat Res 845, 402995. doi:10.1016/j.mrgentox.2018.12.003
Morey, J. S., Ryan, J. C. and Van Dolah, F. M. (2006). Microarray validation: Factors influencing correlation between oligonucleotide microarrays and real-time PCR. Biol Proced Online 8, 175-193. doi:10.1251/bpo126
Parish, S. T., Aschner, M., Casey, W. et al. (2020). An evaluation framework for new approach methodologies (NAMs) for human health safety assessment. Regul Toxicol Pharmacol 112, 104592. doi:10.1016/j.yrtph.2020.104592
SCCNFP (2004). Opinion on Methylisothiazolinone. SCCNFP/0805/04.
SCCP – Scientific Committee on Consumer Products (2005). Opinion on benzoic acid and sodium benzoate. SSCP/0891/05. https://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_015.pdf
SCCP (2006). Opinion on hydroxybenzomorpholine. SCCP/0965/05. https://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_066.pdf
SCCP (2007). Opinion on 4-amino-3-nitrophenol. SCCP/1059/06. https://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_094.pdf
SCCP (2008). Opinion on 1-naphthol. SCCP/1123/07. https://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_125.pdf
SCCS – Scientific Committee on Consumer Safety (2020). Opinion on Dihydroxyacetone (DHA) CAS N° 96-26-4. doi:10.2875/672136
Seo, J. E., Tryndyak, V., Wu, Q. et al. (2019). Quantitative comparison of in vitro genotoxicity between metabolically competent HepaRG cells and HepG2 cells using the high-throughput high-content CometChip assay. Arch Toxicol 93, 1433-1448. doi:10.1007/s00204-019-02406-9
Speit, G., Neuss, S., Schütz, P. et al. (2008). The genotoxic potential of glutaraldehyde in mammalian cells in vitro in comparison with formaldehyde. Mutat Res 649, 146-154. doi:10.1016/j.mrgentox.2007.08.010
Statnikov, A. and Aliferis, C. F. (2007). Are random forests better than support vector machines for microarray-based cancer classification? AMIA Annu Symp Proc 2007, 686-690.
Statnikov, A., Wang, L. and Aliferis, C. F. (2008). A comprehensive comparison of random forests and support vector machines for microarray-based cancer classification. BMC Bioinformatics 9, 319. doi:10.1186/1471-2105-9-319
Tsai, C. C., Huang, R. N., Sung, H. W. et al. (2000). In vitro evaluation of the genotoxicity of a naturally occurring crosslinking agent (genipin) for biologic tissue fixation. J Biomed Mater Res 52, 58-65. doi:10.1002/1097-4636(200010)52:1<58::aid-jbm8>3.0.co;2-0
Vergnes, J. S. and Ballantyne, B. (2002). Genetic toxicology studies with glutaraldehyde. J Appl Toxicol 22, 45-60. doi:10.1002/jat.825
Vo, A. H., Van Vleet, T. R., Gupta, R. R. et al. (2020). An overview of machine learning and big data for drug toxicity evaluation. Chem Res Toxicol 33, 20-37. doi:10.1021/acs.chemrestox.9b00227
Wang, C., Lan, L., Zhang, Y. et al. (2011). Face recognition based on principle component analysis and support vector machine. 3rd International Workshop on Intelligent Systems and Applications, Wuhan, China. doi:10.1109/ISA.2011.5873309
Wei, Y., Tenzen, T. and Ji, H. (2015). Joint analysis of differential gene expression in multiple studies using correlation motifs. Biostatistics 16, 31-46. doi:10.1093/biostatistics/kxu038
Wilson, A. S., Davis, C. D., Williams, D. P. et al. (1996). Characterisation of the toxic metabolite(s) of naphthalene. Toxicology 114, 233-242. doi:10.1016/s0300-483x(96)03515-9
Wu, Y. and Wang, G. (2018). Machine learning based toxicity prediction: From chemical structural description to transcriptome analysis. Int J Mol Sci 19, 2358. doi:10.3390/ijms19082358
Zwiener, I., Frisch, B. and Binder, H. (2014). Transforming RNA-seq data to improve the performance of prognostic gene signatures. PLoS One 9, e85150. doi:10.1371/journal.pone.0085150