Dear readers,
This last issue for 2020 completes my first decade as editor of ALTEX, and I would like to express my heartfelt gratitude to my colleagues, who have all been on the team from the start, for their unfailing enthusiasm and dedication, which made this such a fulfilling and successful time.
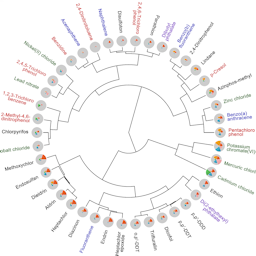
Starting off the issue, Federica Madia and colleagues provide Food for thought … by introducing different approaches aiming to use existing data to predict the results of other regulatory endpoints that would otherwise require animal testing. Their aim is to reduce redundancy and promote and expand the use of non-animal methods for regulatory purposes.
The t4 Workshop Report by Costanza Rovida and co-authors lays out the main issues that need to be addressed for the read-across approach for toxicity assessment to gain better regulatory acceptance worldwide and explains how new approach methodologies can contribute. Biokinetics, which is one of these main issues, is dealt with in more detail in a Consensus Report by Ans Punt and co-authors. Biokinetics can inform on relevant chemical concentrations and metabolites that need to be considered in in vitro tests. The article discusses how this bottleneck can be unblocked by harnessing new technologies but also by forming an OECD expert group on biokinetics to steer the process.
The monocyte activation test was developed to replace pyrogenicity testing in rabbits for parenteral products. Marilena Etna et al. have successfully optimized the test for batch release of tick-borne encephalitis virus vaccines. The assay is shown to be highly sensitive also for this product but requires an adaptation of the validity criteria.
Patients on ventilators are at risk of developing ventilator-induced lung injury (VILI) owing to mechanical stress. Harnessing their human cell model of VILI to investigate the protective effects of azithromycin, which are unrelated to its antibacterial activity, Jon Joelsson and colleagues show that the cell model can be used to elucidate the mechanistic basis of such anti-inflammatory and barrier-protective effects, supporting its relevance as a non-animal model of VILI.
When a bone is fractured, blood cells from ruptured vessels and bone marrow cells together form a fracture hematoma, within which bone healing starts. Moritz Pfeiffenberger et al. present a novel model of the human fracture hematoma, based on primary human cells cultured under hypoxic conditions, with which either drug-induced inhibition of fracture healing or the rescue of delayed fracture healing can be modeled. Thus, this human-relevant method lends itself to the replacement of animal experiments in early preclinical studies of fracture healing.
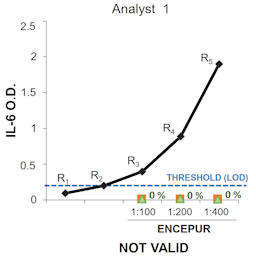
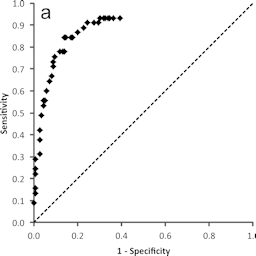
Back to back papers by Britta Wareing et al. and Andreas Natsch et al. report on the kinetic direct peptide reactivity assay (kDPRA), a modification of an OECD test guideline test, which differentiates between strong and weak skin sensitizers. The predictivity of the kDPRA is comprehensively proven based on 180 chemicals in the first paper, and its inter- and intra-laboratory reliability is successfully demonstrated in the second paper. Based on the recommendations of the independent peer review, it is expected that the kDPRA soon will be included in the OECD TG 442C so that it can replace animal tests currently performed for subclassification of skin sensitizers.
Zunwei Chen and colleagues present a high-throughput in vitro screening model to assess the toxicity of environmental chemicals based on a panel of five cell types representing liver, nervous system, heart and endothelium, and a matrix of different endpoints. They find that this model is a conservative and fast alternative to in vivo tests that can deliver essential risk information for decision-making, e.g., in response to chemical spills.
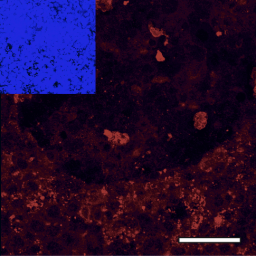
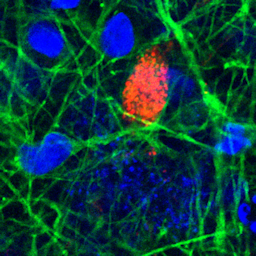
Reports of neurological symptoms in COVID-19 patients raised the question whether the virus can infect the brain. Korin Bullen and team set out to answer this question in the human induced pluripotent stem cell-derived in vitro BrainSphere model, in which they found that the ACE2 receptor is expressed. They show evidence of a productive infection with SARS-CoV-2 in a small population of neural cells, indicating that infection of the brain could endanger vulnerable patient populations.
The BenchMarks article by Jaffar Kisitu explores the difference between nominal and free concentrations of chemicals, what factors affect the free concentration, and how it can be estimated. These considerations are essential for comparison of effective concentrations in different in vitro assays or their extrapolation to other models or applications.
A letter by Katharina Hohlbaum et al. reports on a survey on the views of animal facility and scientific staff on non-aversive mouse handling after a lecture and discussion on the subject. Meeting Reports on ways to increase the availability of human tissue for research purposes, in vitro strategies for food and environmental safety, and COVID-19-related research projects as well as the Corners provide insight into recent 3R-related activities. Please note that current news and upcoming events, including many insightful webinars, are published on our website.
Finally, we wish to thank you, our authors, reviewers, subscribers, readers, members and sponsors, for your continued support of ALTEX in 2020 and hope you and your families stay safe and healthy.
Sonja von Aulock and the ALTEX Edition Editorial Office
with Franz P. Gruber and the Board of ALTEX Edition
Making better use of toxicity studies for human health by extrapolating across endpoints
https://doi.org/10.14573/altex.2005061
The in vitro human fracture hematoma model – a tool for preclinical drug testing
https://doi.org/10.14573/altex.1910211
Internationalization of read-across as a validated new approach method (NAM) for regulatory toxicology
https://doi.org/10.14573/altex.1912181
Increasing the availability of quality human tissue for research
https://doi.org/10.14573/altex.2007141
Innovative in vitro strategies for food and environmental safety
https://doi.org/10.14573/altex.2006261
R2N and the use of alternative methods in COVID-19 research
https://doi.org/10.14573/altex.2010011
Infectability of human BrainSphere neurons suggests neurotropism of SARS-CoV-2
https://doi.org/10.14573/altex.2006111
Chemical concentrations in cell culture compartments (C5) – free concentrations
https://doi.org/10.14573/altex.2008251